
Establishing age-adjusted trabecular bone score curves using dual-energy X-ray absorptiometry in Chinese women and men: a cross-sectional study
Introduction
Osteoporosis is a common age-related disease characterized by low bone mass and subtle changes in bone structure (1). It not only affects the daily life of older adult individuals but also increases the risk of fragility fractures and even death in severe cases (2). The current gold standard for diagnosing and detecting osteoporosis in the clinic is bone mineral density (BMD) as measured by dual-energy X-ray absorptiometry (DXA). BMD and bone mass are commonly regarded as essential parameters for determining fracture risk. The International Society for Clinical Densitometry (ISCD) diagnostic criteria for osteoporosis include a comparison with the BMD reference value for White women aged 20–29 years, called the T-score, with scores required to be within 2.5 standard deviations (SD). Osteopenia refers to a bone density T-score between −1 and −2.5 (3-5). However, some patients with osteoporotic fractures only show osteopenia on BMD measurements, which does not meet the diagnostic criteria for osteoporosis, or the BMD measurement may be within the normal range, suggesting that other factors also affect bone strength (6). Therefore, using the BMD measurement alone is not advised to identify individuals at risk of fractures.
The trabecular bone score (TBS) is a method of analyzing the microstructure of trabecular bone. The TBS analyzes the lumbar spine by extracting texture information from DXA images and uses the principle of pixel difference variables to calculate a final score (7). A high TBS value indicates that the bone microstructure is dense and well-connected, and the space between the spans is small, while a low TBS value indicates a weaker and less fracture-resistant bone microstructure.
Previous reports showed parallel trends in age-related TBS and changes in spine bone density, reflecting the deterioration of bone microstructure with age and menopause (8). This finding suggests that TBS could also be analyzed in the clinic using a specific reference database in a similar manner to BMD. Jain et al. (9) found that the association between fractures and TBS varied according to race/ethnicity and sex when they analyzed data from the US National Health and Nutrition Examination Survey 2005–2008. Therefore, more information is needed to define the appropriate use of TBS for predicting fractures in different populations.
To the best of our knowledge, TBS has only been examined in 1 female population in mainland China, with the study using a Hologic scanner (10). Therefore, we aimed to establish the cutoff points for TBS values in healthy Chinese women and men based on a large population sample, which may serve as the first step to improving the identification of patients at risk of fragility fracture in China. We present the following article in accordance with the STROBE reporting checklist (available at https://qims.amegroups.com/article/view/10.21037/qims-22-550/rc).
Methods
Study population
This retrospective study group included 4,079 unrelated, Han Chinese participants (3,061 women; 1,018 men) aged 20–74 years. Each participant completed a health history questionnaire before a physical examination from July 2018 to October 2021 in the First Affiliated Hospital of Jinan University. We excluded participants who had any of the following: hyperthyroidism, including subclinical and overt hyperthyroidism; diabetes; hyperparathyroidism; renal failure; hypothalamic–pituitary–adrenal diseases; rheumatic diseases; vertebral fracture, vertebroplasty, or degenerative changes with a more than 1 SD BMD from immediately adjacent vertebra; and a BMI that indicated they were underweight [body mass index (BMI) <18 kg/m2] or obese (BMI >30 kg/m2). Two experts independently analyzed all exams. Lumbar spine BMD and TBS were first calculated for vertebrae (L1–4). Finally, participants were selected according to ISCD guidelines, exclusion criteria, and image clarity (Figure 1). The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013) and was approved by the Ethics Committee of the First Affiliated Hospital of Jinan University. All patients signed informed consent forms.
Anthropometric assessments
Anthropometric assessments were conducted with the participants wearing light clothing (gown and shorts) and no shoes. Height and weight were measured using a stadiometer and balance-beam scale, respectively. Body weight was measured to the nearest 0.1 kg, and height was measured to the nearest 0.1 cm (11). BMI was calculated according to the following formula: BMI (kg/m2) = body weight (kg)/height squared (m2).
BMD measurements
We retrospectively analyzed data for consecutive individuals who received total body measurements using DXA at the First Affiliated Hospital of Jinan University from July 2018 to October 2021. BMD measurements were recorded for the lumbar spine (L1–4). All participants were measured using a Lunar iDXA scanner (GE Healthcare, Chicago, IL, USA), which underwent quality control procedures each day before scanning. The scans and analyses were performed and evaluated by the same trained technician. The precision was estimated in 30 volunteers who accepted repeated scanning according to BMD (L1–4) based on the root mean square (RMS) SD values, and the RMS coefficient of variance was <0.7%.
TBS measurements
All TBS measurements were performed at the First Affiliated Hospital of Jinan University using TBS iNsight software (version 3.0.2.0, Med-Imaps, Bordeaux, France). TBS (L1–4) values were calculated using TBS iNsight software for the same anteroposterior spine region used for the lumbar spine BMD measurements. The RMS SD and RMS coefficient of variance for TBS (L1–4) were 0.015 and 0.001, respectively.
Statistical analysis
All analyses were stratified by sex. BMD and TBS values were normally distributed for both men and women. Therefore, means and SDs were calculated for each 5-year interval from 20–74 years. Comparisons between 2 groups were conducted using χ2 tests for percentages and t tests for continuous variables. Pearson correlation coefficients were generated to determine the association between TBS parameters, age, height, weight, BMI, and BMD. Reference plots were created with age (20–74 years) on the x-axis and TBS or BMD on the y-axis. A TBS value greater than 1 SD below the TBS mean of the peak group was considered normal, similar to the ISCD T-score osteoporosis diagnostic criteria under DXA. A TBS 2 SDs below the mean was considered as the cutoff value for low-quality bone in each sex. Values between 1 and 2 SDs below the mean were considered intermediate bone quality, and values higher than 1 SD below the mean were defined as normal bone quality. A P value <0.05 was considered statistically significant.
Results
Baseline characteristics
The overall study population at baseline consisted of 4,079 participants (1,018 men and 3,061 women) aged 20–74 years (Table 1). The mean age of the overall study population was 52.57±13.06 years (men 51.52±13.92 years; women 52.91±12.74 years; P<0.05). Weight, height, and BMI were all significantly higher in men than in women. BMD (L1–4) and TBS (L1–4) were also higher in men than in women (P<0.001). Age was negatively correlated with TBS (L1–4) and BMD (L1–4) in men and in women (P<0.001). TBS (L1–4) showed a significant positive correlation with BMD (L1–4) in both sexes, with the correlation of TBS and BMD being stronger in women than in men (0.78 vs. 0.65; P<0.001).
Table 1
| Variables | Total (n=4,079) | Men (n=1,018) | Women (n=3,061) | P value |
|---|---|---|---|---|
| Age (years) | 52.57±13.06 | 51.52±13.92 | 52.91±12.74 | 0.005 |
| BMI (kg/m2) | 22.90±2.49 | 23.42±2.45 | 22.73±2.47 | <0.001 |
| TBS (L1–4) | 1.34±0.11 | 1.38±0.10 | 1.32±0.10 | <0.001 |
| BMD (L1–4) (g/cm2) | 1.04±0.17 | 1.08±0.16 | 1.02±0.18 | <0.001 |
Data are shown as the mean ± standard deviation or number. BMI, body mass index; TBS, trabecular bone score; BMD, bone mineral density.
Correlation of BMD and TBS of the lumbar spine with age in men and women
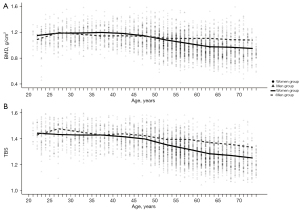
Lumbar spine BMD and TBS decreased with increasing age (Table 2). The mean TBS and lumbar spine BMD decreased from 20–24 years to 70–74 years in both sexes; especially in women after 50 years of age, TBS and spine BMD showed a clear nonlinear decline (Figure 2). The peak age range for TBS (L1–4) and BMD (L1–4) in men was 25–29 years, with a mean peak TBS (L1–4) of 1.47±0.08 and a mean peak BMD (L1–4) of 1.17±0.15 g/cm2. The peak age range for TBS in women was 20–24 years, with a plateau at 20–39 years and a mean peak TBS (L1–4) of 1.43±0.08. The peak age for BMD (L1–4) was 35–39 years, with a mean of 1.17±0.13 g/cm2.
Table 2
| Age (years) | Female | Male | |||||||
|---|---|---|---|---|---|---|---|---|---|
| No. of participants | BMI (kg/m2) | TBS (L1–4) | BMD (L1–4) (g/cm2) | No. of participants | BMI (kg/m2) | TBS (L1–4) | BMD (L1–4) (g/cm2) | ||
| 20–74 | 3,061 | 22.73±2.47 | 1.32±0.10 | 1.02±0.18 | 1,018 | 23.42±2.45 | 1.38±0.10 | 1.08±0.16 | |
| 20–24 | 69 | 21.42±2.76 | 1.43±0.08 | 1.12±0.12 | 37 | 22.94±2.52 | 1.42±0.10 | 1.05±0.21 | |
| 25–29 | 137 | 21.23±2.19 | 1.42±0.09 | 1.16±0.13 | 59 | 23.01±2.53 | 1.47±0.08 | 1.17±0.15 | |
| 30–34 | 162 | 21.60±2.54 | 1.42±0.08 | 1.16±0.12 | 80 | 23.25±2.54 | 1.44±0.08 | 1.14±0.14 | |
| 35–39 | 157 | 22.31±2.63 | 1.42±0.08 | 1.17±0.13 | 53 | 23.62±2.85 | 1.42±0.08 | 1.11±0.13 | |
| 40–44 | 180 | 22.52±2.49 | 1.40±0.07 | 1.15±0.14 | 74 | 23.76±2.53 | 1.43±0.08 | 1.11±0.14 | |
| 45–49 | 305 | 22.70±2.42 | 1.38±0.08 | 1.11±0.16 | 84 | 23.98±2.39 | 1.41±0.10 | 1.10±0.17 | |
| 50–54 | 456 | 22.94±2.37 | 1.33±0.08 | 1.04±0.16 | 130 | 23.74±2.42 | 1.38±0.09 | 1.07±0.16 | |
| 55–59 | 543 | 22.85±2.40 | 1.30±0.08 | 0.98±0.15 | 156 | 23.75±2.32 | 1.38±0.09 | 1.07±0.15 | |
| 60–64 | 482 | 23.02±2.40 | 1.26±0.07 | 0.93±0.13 | 149 | 23.31±2.28 | 1.35±0.08 | 1.07±0.16 | |
| 65–69 | 346 | 23.08±2.33 | 1.24±0.08 | 0.92±0.16 | 120 | 22.79±2.45 | 1.34±0.09 | 1.05±0.18 | |
| 70–74 | 224 | 23.53±2.45 | 1.22±0.07 | 0.90±0.16 | 76 | 23.05±2.30 | 1.31±0.11 | 1.04±0.18 | |
Data are shown as the mean ± standard deviation or number. BMI, body mass index; TBS, trabecular bone score; BMD, mineral density.
Outcomes of TBS cutoff standard references
Given the lack of epidemiological studies on the incidence of osteoporotic fractures in relation to TBS in mainland Chinese populations, the TBS cutoff standard references for the Chinese population should be established. According to the statistical confidence interval, TBS was considered abnormal (bone microarchitecture reduction) if it was 2 SDs below the TBS mean value of the peak TBS group (Table 3).
Table 3
| Sex | Peak age (years) | Mean ± SD of TBS | TBS category (according to SD) |
|---|---|---|---|
| Male | 25–29 | 1.47±0.08 | Normal: TBS ≥1.39 |
| Partially reduced bone microarchitecture: 1.31–1.39 | |||
| Reduced bone microarchitecture: TBS ≤1.31 | |||
| Female | 20–24 | 1.43±0.08 | Normal: TBS ≥1.35 |
| Partially reduced bone microarchitecture: 1.27–1.35 | |||
| Reduced bone microarchitecture: TBS ≤1.27 |
TBS, trabecular bone score; SD, standard deviation.
Discussion
In this study, we analyzed BMD and TBS data for 4,079 participants aged 20–74 years to establish TBS reference values for Chinese men and women. TBS (L1–4) was negatively correlated with age in both men and women. TBS (L1–4) and lumbar spine BMD (L1–4) peaked at the same age (25–29 years) in men, but TBS (L1–4) peaked earlier (20–24 years) than did BMD (35–39 years) in women. There was a significant positive correlation between TBS (L1–4) and lumbar spine BMD (L1–4) in both sexes, similar to the findings of other studies (12,13). However, the correlation was better in women than in men, which might be explained by the higher speed of decrease in bone quality with increasing age in women than in men (14).
The peak age of TBS (L1–4) and BMD (L1–4) in Chinese males was 25–29 years old. Compared with the peak value in Chinese males aged 74 years, TBS and BMD decreased by 10.88% and 11.11%, respectively. In Chinese males aged 50 years, the rate of decline for TBS and BMD from the peak were 5.07% and 2.80%, respectively. One interesting finding was the nonlinear clear decline in TBS and spine BMD with age for women after 50 years of age. The peak TBS occurred at 20–24 years in Chinese women, with a plateau at 20–39 years and a mean peak TBS (L1–4) of 1.43±0.08. In contrast, BMD (L1–4) peaked at 35–39 years, with a mean of 1.17±0.13 g/cm2. A previous study found that TBS (L1–4) peaked at 30.0 years among Iranian men and at 24.5 years in Iranian women, which was consistent with the results of a study in Chinese women using a Hologic DXA device (10,15). In our study, the ages of peak TBS in Chinese women and men were also similar to those in the Iranian population (15). TBS (L1–4) decreased by 14.69%, and BMD (L1–4) decreased by 23.08% in women at 74 years old, compared with the peak value, and by 8.27% and 14.46%, respectively, compared with the values at 50 years old. Compared with women, TBS (L1–4) levels did not plateau in Chinese men between the ages of 20 and 74 years. The declines in TBS and BMD were both greater in women than in men, especially after the age of 50 years, which is related to the decline in estrogen levels after menopause (16).
We followed the method of defining osteoporosis based on BMD, which can be used to detect the peak TBS and its cutoff value of low bone mass. Without long-term follow-up data, this seems to be a reliable method (15). A TBS (L1–4) ≥1.39 was considered normal in Chinese men, with a TBS of 1.31–1.39 indicating partially reduced bone microarchitecture and a TBS ≤1.31 indicating reduced bone microarchitecture. In contrast, a TBS ≥1.35 was considered normal in Chinese women, a score of 1.27–1.35 indicated partial bone microarchitecture reduction, and a score ≤1.27 indicated bone microarchitecture reduction.
Although we still used BMD as the diagnostic criterion for osteoporosis, TBS could also be used as a reference value for low bone mass. Adding the TBS value to existing predicting fracture models, such as FRAX, may improve the accuracy of these models (17). In some studies, TBS value can be a predictor of fracture risk independent of BMD (18). The peak TBS value is often used as the reference standard. In one study, the mean TBS in Canadian men was 1.297±0.107, and the mean in Canadian women was 1.273±0.108 (19), while the equivalent values in Iranian men and women were 1.420±0.094 and 1.428±0.070, respectively (15). Iranians and Chinese are both Asian populations; however, the peak TBS (L1–4) value was lower in the Iranian population compared with that of the Chinese population (15). Furthermore, the peak TBS value in Chinese women was 1.43±0.08 (20–24 years), which was similar to the peak TBS value (1.371±0.066; 20–29 years) in women in another Asian country—Sri Lanka (20). These findings suggest that ethnicity is an important factor affecting bone microstructure, similar to BMD. Therefore, determining the normal range of TBS in the Chinese population would aid in the clinical detection of osteoporosis in China.
The risk of fracture is associated with TBS in many diseases, but there is a lack of TBS criteria for the Chinese population. This study is the first to establish the TBS cutoff point using population data from mainland China. The TBS range can be used to identify the risk difference of osteoporosis among different TBS-level groups in the Chinese population, especially among those with similar BMD t values but large differences in fracture risk (17,21,22).
There were some limitations in our study. To the best of our knowledge, this is the first large cross-sectional study of TBS data in a mainland Chinese population. However, most study participants were from South China, which might limit the application of these TBS reference values in individuals from other parts of China. Furthermore, in our study, a portion of participants who were underweight or overweight or too young or old were excluded. This might have affected the TBS values obtained. Finally, we determined the cutoff based on a peak mean of TBS using a statistical method. For bone health parameters such as TBS, it is better to determine the cutoff based on clinical outcomes such as vertebral fractures that should be determined in a longitudinal study. However, this study did not completely exclude participants with vertebral fractures, such as those with occult fractures, which could not be detected by self-report. Further follow-up studies are required.
In conclusion, our study provides a suitable age-related TBS database and a TBS cutoff standard reference for the Chinese population. If the established cutoff point is confirmed in a subsequent prospective study, this database will improve the clinical understanding of changes in patients’ bone microstructure and aid in the diagnosis of osteoporosis and prediction of fracture risk in Chinese individuals.
Acknowledgments
Funding: This study was financially supported by the National Natural Science Foundation of China (No. 81871383).
Footnote
Reporting Checklist: The authors have completed the STROBE reporting checklist. Available at https://qims.amegroups.com/article/view/10.21037/qims-22-550/rc
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://qims.amegroups.com/article/view/10.21037/qims-22-550/coif). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013) and was approved by the institutional review board of the First Affiliated Hospital, Jinan University. Informed consent was obtained from all the patients.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Noel SE, Santos MP, Wright NC. Racial and Ethnic Disparities in Bone Health and Outcomes in the United States. J Bone Miner Res 2021;36:1881-905. [Crossref] [PubMed]
- Beaudart C, Hiligsmann M, Li N, Lewiecki EM, Silverman S. Effective communication regarding risk of fracture for individuals at risk of fragility fracture: a scoping review. Osteoporos Int 2022;33:13-26. [Crossref] [PubMed]
- Akid I, Doberman DJ. Bone Health. Clin Geriatr Med 2021;37:683-96. [Crossref] [PubMed]
- Ulivieri FM, Rinaudo L. Beyond Bone Mineral Density: A New Dual X-Ray Absorptiometry Index of Bone Strength to Predict Fragility Fractures, the Bone Strain Index. Front Med (Lausanne) 2021;7:590139. [Crossref] [PubMed]
- Hans D, Downs RW Jr, Duboeuf F, Greenspan S, Jankowski LG, Kiebzak GM, Petak SM. Skeletal sites for osteoporosis diagnosis: the 2005 ISCD Official Positions. J Clin Densitom 2006;9:15-21. [Crossref] [PubMed]
- Shevroja E, Lamy O, Kohlmeier L, Koromani F, Rivadeneira F, Hans D. Use of Trabecular Bone Score (TBS) as a Complementary Approach to Dual-energy X-ray Absorptiometry (DXA) for Fracture Risk Assessment in Clinical Practice. J Clin Densitom 2017;20:334-45. [Crossref] [PubMed]
- Löffler MT, Sollmann N, Mei K, Valentinitsch A, Noël PB, Kirschke JS, Baum T. X-ray-based quantitative osteoporosis imaging at the spine. Osteoporos Int 2020;31:233-50. [Crossref] [PubMed]
- Martineau P, Silva BC, Leslie WD. Utility of trabecular bone score in the evaluation of osteoporosis. Curr Opin Endocrinol Diabetes Obes 2017;24:402-10. [Crossref] [PubMed]
- Jain RK, Vokes T. Association of trabecular bone score (TBS) and prior fracture differs among minorities in NHANES 2005-2008. Osteoporos Int 2018;29:2093-9. [Crossref] [PubMed]
- Cheng P, Qi HM, Di WJ, Liu J, Yu J, Lv S, Shen YL, Zha JM, Cai JM, Lai B, Ding GX. Establishment of TBS reference plots and correlation between TBS and BMD in healthy mainland Chinese women. Arch Osteoporos 2016;11:5. [Crossref] [PubMed]
- Ke D, Lu D, Cai G, Wang X, Zhang J, Suzuki K. Chronological and Skeletal Age in Relation to Physical Fitness Performance in Preschool Children. Front Pediatr 2021;9:641353. [Crossref] [PubMed]
- Krohn K, Schwartz EN, Chung YS, Lewiecki EM. Dual-energy X-ray Absorptiometry Monitoring with Trabecular Bone Score: 2019 ISCD Official Position. J Clin Densitom 2019;22:501-5. [Crossref] [PubMed]
- Dufour R, Winzenrieth R, Heraud A, Hans D, Mehsen N. Generation and validation of a normative, age-specific reference curve for lumbar spine trabecular bone score (TBS) in French women. Osteoporos Int 2013;24:2837-46. [Crossref] [PubMed]
- Iki M, Tamaki J, Sato Y, Winzenrieth R, Kagamimori S, Kagawa Y, Yoneshima H. Age-related normative values of trabecular bone score (TBS) for Japanese women: the Japanese Population-based Osteoporosis (JPOS) study. Osteoporos Int 2015;26:245-52. [Crossref] [PubMed]
- Shafiee G, Sharifi F, Heshmat R, Ostovar A, Ebrahimpur M, Sheidaei A, Nabipour I, Larijani B. The reference value of trabecular bone score (TBS) in the Iranian population. J Diabetes Metab Disord 2020;19:493-8. [Crossref] [PubMed]
- Huang ML, Hsieh TJ, Lin SS, Huang WC. Spine trabecular bone scores and bone mineral density of postmenopausal Taiwanese women. Menopause 2022;29:1308-14. [Crossref] [PubMed]
- McCloskey EV, Odén A, Harvey NC, Leslie WD, Hans D, Johansson H, et al. A Meta-Analysis of Trabecular Bone Score in Fracture Risk Prediction and Its Relationship to FRAX. J Bone Miner Res 2016;31:940-8. [Crossref] [PubMed]
- Buehring B, Thomas J, Wittkämper T, Baraliakos X, Braun J. Evaluation of the trabecular bone score (TBS) in routine clinical care of patients with inflammatory rheumatic and non-inflammatory diseases: Correlation with conventional bone mineral density measurement and prevalence of vertebral fractures. Z Rheumatol 2020;79:1067-74. [Crossref] [PubMed]
- Mazzetti G, Berger C, Leslie WD, Hans D, Langsetmo L, Hanley DA, Kovacs CS, Prior JC, Kaiser SM, Davison KS, Josse R, Papaioannou A, Adachi JR, Goltzman D, Morin SN. Densitometer-Specific Differences in the Correlation Between Body Mass Index and Lumbar Spine Trabecular Bone Score. J Clin Densitom 2017;20:233-8. [Crossref] [PubMed]
- Rathnayake H, Lekamwasam S, Wickramatilake C, Lenora J. Trabecular bone score and bone mineral density reference data for women aged 20-70 years and the effect of local reference data on the prevalence of postmenopausal osteoporosis: a cross-sectional study from Sri Lanka. Arch Osteoporos 2019;14:91. [Crossref] [PubMed]
- Iki M, Tamaki J, Kadowaki E, Sato Y, Dongmei N, Winzenrieth R, Kagamimori S, Kagawa Y, Yoneshima H. Trabecular bone score (TBS) predicts vertebral fractures in Japanese women over 10 years independently of bone density and prevalent vertebral deformity: the Japanese Population-Based Osteoporosis (JPOS) cohort study. J Bone Miner Res 2014;29:399-407. [Crossref] [PubMed]
- Lee JE, Kim KM, Kim LK, Kim KY, Oh TJ, Moon JH, Choi SH, Lim S, Kim SW, Shin CS, Jang HC. Comparisons of TBS and lumbar spine BMD in the associations with vertebral fractures according to the T-scores: A cross-sectional observation. Bone 2017;105:269-75. [Crossref] [PubMed]



