
Simultaneous dual-plane, real-time magnetic resonance imaging of oral cavity movements in advanced trombone players
Introduction
Real-time magnetic resonance imaging has been utilized as a method for obtaining descriptive and quantitative kinematic information in a wide variety of applications. Among these are cardiac dynamics (1-4), speech (5), swallowing (6), and oral cavity movements during brass instrument performance (7-12).
With respect to these studies of brass instrumentalists, the most commonly utilized vantage point has been the mid-sagittal plane, with qualitative and quantitative analyses being achieved by creating profile lines at different locations within the oral cavity. Temporal changes in pixel luminescence along these profile lines as a function of movements of the tongue are then measured to quantify very fast tongue movements (9,13), and allow for spatial and temporal comparisons between advanced, healthy (AH) French horn players and those affected by embouchure dystonia (EmD) (7,8).
However, there are structures within the oral cavity and throat of brass musicians that cannot be readily observed from sagittal images, requiring alternate views. For example, the glottis has only recently been shown to be intricately involved in regulating loudness and pitch in AH French horn players by studying real-time MRI images captured in the coronal plane (10). Additionally, an airway channel has been identified in these musicians which is formed between the dorsal surface of the tongue and the hard palate that also modifies its dimensions during French horn performance. These findings demonstrate differences between AH players and those with EmD when playing sustained notes (14), as well as differences that vary with loudness and pitch in AH players (10).
Previous studies have shown that movements of the tongue occurring in French horn players are seen during the performance of specific exercises that require the performer to play different notes at different loudness levels (7-12). The extent to which these movements are coordinated in time is presently unknown, however, such information may be useful to brass students and teachers if coincident movement contributes to successful performance. It would be possible to estimate such coordination by having players perform repeated trials of the same exercise, one time imaging the structure under consideration in the sagittal plane, and on a separate trial viewing it in a different (e.g., transverse or coronal) plane. By aligning such movements to audio tracks obtained during filming, it would be possible to approximate the degree of temporal synchrony. However, the reliability of such an approach is less than optimal, as significant and unknown inter-trial variations in movements might exist that could mask the true relationship.
This work evaluates a new application of real-time MRI technology by which images are obtained in two perpendicular planes (15) simultaneously during the performance of a musical exercise, designed to elicit significant movement of the tongue, in AH trombone players. This approach, for the first time, allows the precise determination of the degree of temporal synchrony that exists between movements of different areas of the tongue in two orthogonal planes, i.e., in three directions.
Methods
Subjects, performance device and testing procedure
Five AH trombone players (age range, 26–48) volunteered to participate as subjects for this study. Two were professional trombonists performing in major German ensembles, and three were musical conservatory students in Germany. Prior to MRI examination, all subjects gave written informed consent as approved by the recommendations of the local ethics committee.
All testing sessions were conducted at the Max-Planck-Institute for Biophysical Chemistry, Göttingen, Germany. For each testing session, the subjects were asked to perform a musical exercise consisting of a series of slurred, ascending and descending intervals spanning two complete octaves (see Figure 1). These intervals, ranging from Bb2 to Bb4, comprise the majority of the natural harmonic series for the trombone. The trombone used was a custom built, non-ferromagnetic instrument designed specifically for MRI studies (Yamaha Corporation of America, Buena Park, CA, USA). Prior to testing, the subjects were given a warm-up/trial period to become familiar with the instrument and with the exercise.
For testing, each subject assumed the supine position. The bottom, bell portion of the trombone was secured at the front end of the scanner table near the subject’s feet, and semi-flexible plastic tubing of the appropriate length and diameter for a trombone was used to extend the tubing up into the magnet and to the subject’s head. A plastic mouthpiece was attached at this end which the subject was able to position appropriately to assume a comfortable playing position. Additionally, a fiber-optic microphone (Dual Channel-FOMRI, Optoacoustics, Or Yehuda, Israel) was secured to the bell of the instrument to record sound. Each subject was granted time to practice in this position until they felt ready to begin. A video monitor was positioned opposite the magnet bore which was visible to the performer by use of a mirror attached to the receiving head coil. During testing, an image of the musical exercise was projected thereon. In addition, a circular LED array was positioned next to the monitor in the window of the scanner room that was also visible to the musician and which flashed at the prescribed tempo for the exercise to assist the performer in keeping time.
Dual-slice real-time MRI
All experiments were performed on a 3 T MRI system (Magnetom Prisma, Siemens Healthineers, Erlangen, Germany) with a maximum gradient strength of 80 mT·m−1. Data acquisition was accomplished with use of a 64-channel head coil. Real-time MRI as originally described in (16), employed highly undersampled radial FLASH acquisitions with randomized radiofrequency spoiling (17). The reconstruction of dynamic images and corresponding coil sensitivity maps defines a nonlinear inverse problem which was solved by the iteratively regularized Gauss-Newton method (16). High acceleration factors were achieved by using complementary sets of radial spokes in successive frames and by temporal regularization of the iterative optimization of the actual frame to the immediately preceding image (and its coil sensitivities). To ensure a high degree of temporal fidelity all regularization terms are downsized by a factor of two during each Gauss-Newton iteration (typically 6 steps). The accuracy of this strategy has been experimentally confirmed (18). For similar reasons, simultaneous dual-slice acquisitions (15) were realized in a frame-interleaved manner which keeps the temporal footprint short (i.e., here at 20 ms), whereas alternative spoke-interleaved acquisitions double the repetition interval and therefore also the measuring time. The chosen strategy further benefits from an improved signal-to-noise ratio due to the interleaved periods of free T1 relaxation. Finally, post-processing of the reconstructed images involved the application of a temporal median filter to reduce residual streaking artifacts and denoising with use of a modified non-local means filter (19).
All real-time MRI measurements were performed with an in-plane resolution of 1.4 mm, slice thickness of 8 mm, and field-of-view of 192×192 mm2. Acquisitions at 2×25 fps employed the following parameters: TR =2.22 ms, TE =1.33 ms (gradient asymmetry =0.4), flip angle = 5°, and 9 radial spokes per image yielding an acquisition time of 20 ms per frame. Online reconstruction and display of real-time images with minimal delay was achieved by a parallelized version of the algorithm (20) and a computer (sysGen, Bremen, Germany) which was fully integrated into the reconstruction pipeline of the commercial MRI system and equipped with 8 graphical processing units (GPUs, GeForce GTX, TITAN, NVIDIA, Santa Clara, CA, USA).
Synchronization of image and audio recordings was achieved by use of TTL trigger signals which were generated by the actual MRI acquisition marking the exact beginning and end of imaging, respectively. The TTL signal is converted into an audio signal (Dual Channel-FOMRI) and recorded along with the audio track. During post-processing the audio signal can then correctly be assigned to the corresponding image series.
Figure 2 provides a still image taken from one of the dual-slice movies.
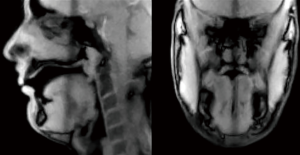
Data analysis
For conducting quantitative analyses, the dual-slice movies were studied separately in the two orientations, each with correctly aligned audio tracks. Because the interleaved, dual-slice films had a 20 ms offset between sagittal and coronal images, this very minor offset was preserved in the resulting movies that were analyzed.
The methods for obtaining quantitative information from real-time MRI films, as well as specific applications in brass musicians have been previously reported (7-11,13,14,21). We employed the same custom real-time MRI toolbox developed for MATLAB (MATLAB R2014a, including the Image Processing and Signal Processing Toolboxes) as in previous research, however, we only report on the methods that are unique to the current study.
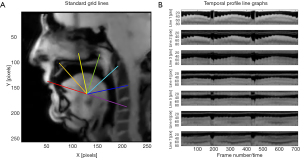
For sagittal views, a set of grid lines was created at 30° intervals from a hand-digitized, standardized baseline using the tip of the front incisor as one landmark and the inferior-anterior edge of the 2nd inter-vertebral disc as the other (see Figure 3A). Based on these grid lines, temporal profile line graphs depicting movements of the tongue edge along the baseline as well as along each grid line were generated in MATLAB, allowing the visualization and calculation of changes in position with respect to time (see Figure 3B).
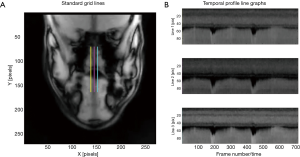
For the simultaneously-acquired coronal views, three grid lines were manually positioned for each subject. These were chosen in order to be able to track vertical movements of the dorsal tongue surface (DTS) in different locations. This yielded an indication of any asymmetry of movement across the width of the tongue as it forms an air channel in combination with the hard palate above. In Figure 4A, the channel formed between the tongue surface and the hard palate above is easily identified, and for these analyses, one vertical grid line was placed in the center of the nose, with the other two placed approximately equidistant and laterally to the right and left. The three resulting profile line graphs are shown in Figure 4B.
Utilizing the customized real-time MRI toolkit, we located the position of the DTS along each profile line for every frame in both the sagittal and coronal views. Using the PRAAT software (22), the audio tracks were automatically segmented based on silences occurring between notes, and manually checked before the time stamps of the onset, midpoint, and ending of each note were determined by the software. By examining signal intensity graphs for each note, we determined that the notes were most stable during a 400 ms window centered on the midpoint of each note. The timing and frame numbers corresponding to this window were measured, and tongue position values (pixels) during that time were averaged to represent a single mean value for each note. Because there were 33 notes in this exercise (see Figure 1), this resulted in 33 values. These values were subsequently averaged across all 7 subjects yielding a single mean tongue position for every note for each profile line. By examining patterns contained in the resulting graphs from these data, direct comparisons between sagittal and coronal views were possible. Finally, we determined the degree of movement concordance between the sagittal and coronal views by calculating cross-correlations were calculated using SPSS (SPSS, Version 25). More specifically, the cross-correlation of the mean DTS positions at each 400 ms window during the exercise was calculated for each different combination of sagittal and coronal profile lines (see Table 1).

Full table
Results
Sagittal images
Examination of Figure 3B indicates a consistent pattern of tongue movement along the first four profile lines (1-4) only, hence only these profile lines were selected for subsequent analyses. Previous models of motor control of the tongue have identified at least two functionally distinct regions of movement in terms of its sagittal orientation (23) with a more recent paper indicating that this division is probably more fine-grained and fluid, allowing the coupling or separation of various tongue segments across different tongue movements (24). The patterns observed in our subject data occur in three distinct regions, each comprised of about 200 frames (i.e., 8,000 ms). Each region corresponds to one of the three note sequences depicted in Figure 1. In each of the regions delineated by the four profile lines, the tongue assumes a retracted, low position in the mouth on the low starting note in each sequence (Bb2), and moves progressively upward along each profile line as the note sequence ascends. As the midpoint of each region corresponds to progressively higher-pitched notes (F4, Ab4, and Bb4, respectively), the position of the DTS in each sequence can be observed to move increasingly further away from the baseline. These patterns were similar among all subjects, and are in agreement with our previous work studying French horn players (7,8). Our current findings regarding trombone players thus confirm that there is a constriction of the air channel formed above the tongue during ascending note sequences, and a widening of the channel during descending note sequences.
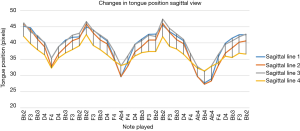
Figure 5 depicts the mean vertical position of the DTS along the four profile lines selected from Figure 4B (lines 1–4, starting from the top) across all subjects during the 400 ms window (described above) as seen in the sagittal plane for all 33 notes in the exercise. Breathing periods between note sequences are not shown. Note that movements of the DTS seen in Figure 3B actually indicate movements from higher pixel vales to lower values during the ascending note phase of each sequence while the opposite is true during the descending phase. Hence, Figure 5 is a reverse image of Figure 3B, comprised only of the mean position of each 400 ms period. Each negative slope in Figure 5 corresponds to an ascending note sequence, and each positive slope corresponds to a descending note sequence. As the exercise consists of three distinct ascending/descending sequences, there are three separate nadirs that progressively get deeper as the top note in each sequence is higher than its predecessor. This corresponds to progressively larger excursions of the tongue along each profile line with each successive sequence (10.9, 13.7, and 15.8 pixels for sequences 1, 2, and 3, respectively).
Coronal images
The subject represented in the coronal view depicted in Figure 4A shows a degree of tongue asymmetry, with the right side of the DTS higher than the left in the depicted, static position. Although we did not observe tongue asymmetries in all subjects, speech production research indicates that they occur quite commonly (25,26). Apart from these individual differences, the movement patterns depicted in Figure 4B were consistent across subjects. Figure 4 overall presents similar findings to those seen in the sagittal view, although the DTS movements imaged in the coronal plane all occur vertically at three different locations across the tongue surface. Specifically, during ascending note sequences, the DTS moves upward, and during descending sequences, it moves downward.
In examining the dark areas above the DTS in each of the profile line graphs of Figure 4B, a second pattern becomes evident. Profile line 2, tracking temporal changes in mid-DTS position, indicates that this region is consistently positioned further from the hard palate, than profile lines 1 and 3 (right-DTS and left-DTS, respectively, although the movement trajectories are overall similar. In other words, the degree of constriction of the air channel during ascending notes is greater at the lateral borders of the tongue than in the middle. For example, during the production of the top note in the third note sequence (Bb4), the lateral borders of the DTS are shown to be in actual contact with the hard palate, as the dark region between the DTS and the hard palate no longer is visible. A recent publication on speech production using electropalatography which measures tongue contact with the rigid structures on the top of the mouth including the inner edge of the upper dentition, showed that lateral tongue contact or bracing against the molars occurred during 97.5% of the total recording duration for the two speakers investigated in the study (26). Given that brass playing requires significantly more airflow and the precise channeling of the air stream to the embouchure, it can be assumed that such bracing also occurs during brass instrument performance (27). Tongue bracing furthermore helps explain how the tongue (described as a muscular hydrostat) can move flexibly within the oral cavity with the help of few extrinsic muscles and in absence of clear antagonistic muscle pairs (26).
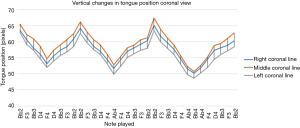
Figure 6 depicts the mean vertical position of the DTS across all subjects during the 400 ms sampling period of each note in the exercise. The contours of this graph may be interpreted in the same way as Figure 5. Specifically, downward movements of the line indicate upward vertical movements of the DTS occurring during ascending notes, and upward movements of the line indicate downward vertical movements of the DTS during descending notes. The position of the DTS along the middle profile line shows consistently larger pixel distances than along either of the lateral profile lines. This indicates that the middle-DTS has not moved up as far as in the other two locations, creating a wider air channel at that position (see Figure 4). This relationship is sustained throughout the entire exercise.
Concurrency of sagittal and coronal movements
The pattern of movement in the coronal plane depicted in Figure 6 closely corresponds to the pattern seen in the sagittal plane in Figure 5. The peaks and valleys occur approximately at the same time in both graphs, implying tight spatial and temporal coupling in both planes. The results of the cross-correlational analysis of the pooled sagittal and coronal DTS positions are listed in Table 1.
The correlation values range from 0.937–0.990, indicating a very strong coupling of DTS movements along any given pairing of coronal and sagittal profile lines. In anatomical terms, this is to be expected, as both planes of observation cover the same tongue. However, movements of the tongue are very complex, requiring involvement of no fewer than 4 paired intrinsic muscles and 4 extrinsic muscles (28). Sagittal observations of tongue movements, being only two-dimensional, fail to show shape changes that may occur across the tongue’s medial-lateral extent. For example, Figure 4A shows a distinct depression in the DTS in the middle relative to the lateral aspects of the tongue. This is shown in Figure 4B as a greater space between the middle-DTS and the hard palate in this location when compared to either the left or right DTS position. This depression can only be created by differential activation of specific intrinsic muscles of the tongue across its width. All three coronal profile lines demonstrate coordinated movements that appear to sustain this depression throughout the exercise, and these are closely linked to movements in the sagittal plane occurring at the same time. The full extent of these tightly coordinated muscle movements can only be discerned using dual-slice acquisition.
Conclusions
This preliminary work demonstrates the feasibility and utility of dual-slice real-time MRI movies in qualitatively and quantitatively describing movements of the tongue during trombone performance. By eliminating the need of recording repeated exercise trials in different planes successively, a more precise estimation of movement coordination can be achieved. Future studies should attempt to use this technology to examine more anatomically discrete structures. For example, in brass musicians, measurements of the linkage between movements of the vocal folds comprising the glottis have been previously reported using repeated exercise trials (10). Although a close coupling appeared to exist between the observed movements of the glottis and movements of the tongue in successive trials of the same exercise, dual-slice acquisition would verify this. Certainly other applications related to speech and cardiovascular function could also benefit from the application of this technology.
Acknowledgments
We wish to acknowledge the contributions of Mr. Douglas Yeo and the Yamaha Corporation for their development of the MRI-compatible trombone used in this study.
Funding: M Heyne received a travel grant from Boston University’s Dudley Allen Sargent Research Fund, and his project contributions were also supported by funding from the NIH (research grant R01DC002852, Principal Investigator Frank Guenther).
Footnote
Conflicts of Interest: J Frahm holds a patent about the real-time MRI acquisition and reconstruction technique used here. The other authors have no conflicts of interest to declare.
Ethical Statement: This study was approved by the recommendations of the local ethics committee, and all subjects gave written informed consent.
References
- Joseph A, Kowallick JT, Merboldt KD, Voit D, Schaetz S, Zhang S, Sohns JM, Lotz J, Frahm J. Real-time flow MRI of the aorta at a resolution of 40 msec. J Magn Reson Imaging 2014;40:206-13. [Crossref] [PubMed]
- Joseph AA, Merboldt KD, Voit D, Zhang S, Uecker M, Lotz J, Frahm J. Real-time phase-contrast MRI of cardiovascular blood flow using undersampled radial fast low-angle shot and nonlinear inverse reconstruction. NMR Biomed 2012;25:917-24. [Crossref] [PubMed]
- Voit D, Zhang S, Unterberg-Buchwald C, Sohns JM, Lotz J, Frahm J. Real-time cardiovascular magnetic resonance at 1.5 T using balanced SSFP and 40 ms resolution. J Cardiovasc Magn Reson 2013;15:79. [Crossref] [PubMed]
- Zhang S, Joseph AA, Voit D, Schaetz S, Merboldt KD, Unterberg-Buchwald C, Hennemuth A, Lotz J, Frahm J. Real-time magnetic resonance imaging of cardiac function and flow-recent progress. Quant Imaging Med Surg 2014;4:313-29. [PubMed]
- Niebergall A, Zhang S, Kunay E, Keydana G, Job M, Uecker M, Frahm J. Real-time MRI of speaking at a resolution of 33 ms: undersampled radial FLASH with nonlinear inverse reconstruction. Magn Reson Med 2013;69:477-85. [Crossref] [PubMed]
- Zhang S, Olthoff A, Frahm J. Real-time magnetic resonance imaging of normal swallowing. J Magn Reson Imaging 2012;35:1372-9. [Crossref] [PubMed]
- Iltis PW, Frahm J, Voit D, Joseph A, Burke R, Altenmüller E. Inefficiencies in motor strategies of horplayers with embouchure dystonia. Med Probl Perform Art 2016;31:69-77. [Crossref] [PubMed]
- Iltis PW, Frahm J, Voit D, Joseph A, Schoonderwaldt E, Altenmüller E. Divergent oral cavity motor strategies between healthy elite and dystonic horn players. J Clin Mov Disord 2015;2:15. [Crossref] [PubMed]
- Iltis PW, Frahm J, Voit D, Joseph AA, Schoonderwaldt E, Altenmüller E. High-speed real-time magnetic resonance imaging of fast tongue movements in elite horn players. Quant Imaging Med Surg 2015;5:374-81. [PubMed]
- Iltis PW, Gillespie SL, Frahm J, Voit D, Joseph A, Altenmüller E. Movements of the Glottis During Horn Performance: A Pilot Study. Med Probl Perform Art 2017;32:33-9. [Crossref] [PubMed]
- Iltis PW, Schoonderwaldt E, Zhang S, Frahm J, Altenmüller E. Real-time MRI comparisons of brass players: A methodological pilot study. Hum Mov Sci 2015;42:132-45. [Crossref] [PubMed]
- Schumacher M, Schmoor C, Plog A, Schwarzwald R, Taschner C, Echternach M, Richter B, Spahn C. Motor functions in trumpet playing-a real-time MRI analysis. Neuroradiology 2013;55:1171-81. [Crossref] [PubMed]
- Iltis PW, Frahm J, Voit D, Joseph A, Altenmüller E, Miller A. Movements of the Tongue during Lip Trills in Horn Players: Real-Time MRI Insights. Med Probl Perform Art 2017;32:209-14. [Crossref] [PubMed]
- Iltis PW, Frahm J, Altenmüller E, Voit D, Joseph A, Kozakowski K. Tongue Position Variability During Sustained Notes in Healthy vs Dystonic Horn Players Using Real-Time MRI. Med Probl Perform Art 2019;34:33-8. [Crossref] [PubMed]
- Krohn S, Joseph AA, Voit D, Michaelis T, Merboldt KD, Buergers R, Frahm J. Multi-slice real-time MRI of temporomandibular joint dynamics. Dentomaxillofac Radiol 2018. Epub ahead of print. [Crossref] [PubMed]
- Uecker M, Zhang S, Voit D, Karaus A, Merboldt KD, Frahm J. Real-time MRI at a resolution of 20 ms. NMR Biomed 2010;23:986-94. [Crossref] [PubMed]
- Roeloffs V, Voit D, Frahm J. Spoiling without additional gradients: Radial FLASH MRI with randomized radiofrequency phases. Magn Reson Med 2016;75:2094-9. [Crossref] [PubMed]
- Klosowski J, Frahm J. Image denoising for real-time MRI. Magn Reson Med 2017;77:1340-52. [Crossref] [PubMed]
- Frahm J, Schaetz S, Untenberger M, Zhang S, Voit D, Merbolt KD. On the Temporal Fidelity of Nonlinear Inverse Reconstructions for Real- Time MRI – The Motion Challenge. Open Med Imaging J 2014;8:1-7. [Crossref]
- Schaetz S, Voit D, Frahm J, Uecker M. Accelerated Computing in Magnetic Resonance Imaging: Real-Time Imaging Using Nonlinear Inverse Reconstruction. Comput Math Methods Med 2017;2017:3527269. [Crossref] [PubMed]
- Altenmüller E, Baur V, Hofmann A, Lim VK, Jabusch HC. Musician's cramp as manifestation of maladaptive brain plasticity: arguments from instrumental differences. Ann N Y Acad Sci 2012;1252:259-65. [Crossref] [PubMed]
- Boersma P. Praat, a system for doing phonetics by computer. Glot International 2001;5:341-5.
- Miyawaki K, Hirose H, Ushimima T, Sawashima M. A prelminary report on the electromyographic study of the activity of lungual muscles. Ann Bull RILP 1975;9:406.
- Stone M, Epstein MA, Iskarous K. Functional segments in tongue movement. Clin Linguist Phon 2004;18:507-21. [Crossref] [PubMed]
- Cheng L, Schellenberg M, Gick B. Cross-Linguistic bracing: a lingual ultrasound study of six languages. Canadian Acoustics 2017;45:186-7.
- Gick B, Allen B, Roewer-Despres F, Stavness I. Speaking Tongues Are Actively Braced. J Speech Lang Hear Res 2017;60:494-506. [Crossref] [PubMed]
- Hartel K. In aller Munde: Die Zunge. Clarino 2012;2:20-3.
- Sanders I, Mu L. A three-dimensional atlas of human tongue muscles. Anat Rec (Hoboken) 2013;296:1102-14. [Crossref] [PubMed]



