The association of neutrophil-lymphocyte ratio with stroke recurrence in patients with symptomatic nonacute atherosclerotic internal carotid artery occlusion
Introduction
Atherosclerotic internal carotid artery occlusion (AICAO) is a major contributor to the emergence of cerebrovascular events, accounting for approximately 10% of transient ischemic attacks (TIAs) and 20% of ischemic strokes (1,2). For patients with nonacute AICAO, the primary treatment approach involves aggressive medical management, including antiplatelet therapy and intensive control of vascular risk factors, such as hypertension, diabetes mellitus, and hyperlipidemia. Surgical treatment for symptomatic nonacute AICAO has not demonstrated superiority over medical management (3,4). However, despite optimal medical treatment, the stroke recurrence rate remains high, ranging from 12.3% to 22.7% within 2 years (3,4). Presently, there are few accurate predictors of stroke recurrence in patients with symptomatic nonacute AICAO (2,5).
Atherosclerosis is a lipid-driven inflammatory process, with inflammation playing a crucial role in the onset and progression of atherosclerosis and its complications (6). The neutrophil-lymphocyte ratio (NLR), calculated as the ratio of neutrophils to lymphocytes measured in peripheral blood, reflects the balance between acute and chronic inflammation and adaptive immunity (7). In recent years, there has been growing interest in identifying easily accessible and cost-effective inflammatory markers that can predict adverse outcomes in cardiovascular and cerebrovascular diseases. Among these, the NLR has emerged as a promising biomarker (8,9). Studies have also demonstrated a higher stroke recurrence rate in patients with an elevated NLR who have experienced acute ischemic stroke (10,11), and among these patients, the NLR is an independent predictor of short-term mortality (12-14). However, these studies primarily involved patients with acute stroke, and the predictive value of NLR for stroke recurrence in patients with nonacute AICAO has not been thoroughly investigated. This study thus aimed to examine the value of NLR in predicting stroke recurrence in patients with symptomatic nonacute AICAO. We present this article in accordance with the STROBE reporting checklist (available at https://qims.amegroups.com/article/view/10.21037/qims-2025-1437/rc).
Methods
Study population
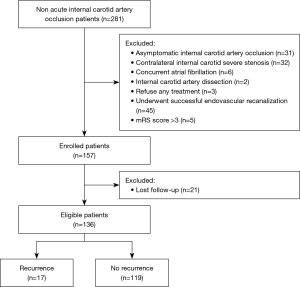
From January 2021 to April 2023, this study retrospectively reviewed patients diagnosed with internal carotid artery occlusion by computed tomography angiography (CTA), magnetic resonance angiography (MRA), or digital subtraction angiography (DSA) at Beijing Tiantan Hospital. Patients were eligible for inclusion if they met the following criteria: age ≥18 years, symptomatic nonacute AICAO occurring more than 30 days from the last qualifying event, and administration of aggressive medical treatments. Meanwhile, the exclusion criteria included the following: acute internal carotid artery occlusion, asymptomatic internal carotid artery occlusion, presence of contralateral internal carotid severe stenosis >70%, history of atrial fibrillation, internal carotid artery occlusion of a nonatherosclerotic etiology (e.g., vasculitis or moyamoya disease), refusal of any treatment, successful revascularization, loss to follow-up, infectious disease arising within 2 weeks before enrollment were excluded, and a baseline modified Rankin Scale (mRS) score >3 (15). This study was conducted in accordance with the Declaration of Helsinki and its subsequent amendments and was approved by the Institutional Review Board of Beijing Tiantan Hospital (No. KY2022-077-02). Informed consent was obtained from all participants.
Laboratory analysis
Peripheral blood samples were collected from the antecubital vein in all enrolled patients at least 14 days after the last qualifying event and within 30 days of the radiological examination. An automatic cell counter was used to analyze the blood samples. Blood samples were obtained at least 14 days after the last qualifying event to minimize transient inflammatory changes. The NLR was calculated by dividing the neutrophil count by the lymphocyte count (16).
Aggressive medical treatment
Patients were prescribed dual-antiplatelet therapy (DAPT), consisting of aspirin (100 mg daily) plus either clopidogrel (75 mg daily) or ticagrelor (90 mg twice daily) for 90 days from symptom onset. Subsequently, they were transitioned to single-antiplatelet therapy with either aspirin, clopidogrel, or ticagrelor based on the results of the platelet aggregation test.
Risk factor management targets were based on the American Heart Association/American Stroke Association (AHA/ASA) guidelines (17) and included systolic blood pressure ≤140 mmHg, glycosylated hemoglobin (HbA1c) ≤7%, and low-density lipoprotein (LDL) level ≤70 mg/dl. Smoking cessation was also strongly encouraged. Stroke neurologists and specialist physicians managed hypertension, diabetes, and hypercholesterolemia treatment. Dietitians and stroke nurses provided education on smoking cessation, diet, and lifestyle modifications.
Clinical characteristics and outcomes
Data collected included patient demographics, past medical history, laboratory studies, radiologic imaging studies [including DSA, computed tomography (CT), and magnetic resonance imaging (MRI)], medications, risk management factors control status, and stroke recurrence. The primary outcome was ipsilateral recurrent stroke during follow-up, as defined by the AHA/ASA criteria (18). The primary outcome events were followed up either in the clinic or by telephone. Patients with blood pressure, LDL, and blood glucose levels meeting target control values, along with successful smoking cessation, were considered to have controlled risk factors.
Statistical analysis
Continuous variables are presented as the median and interquartile range (IQR), while categorical variables are presented as numbers and percentages. Clinical characteristics were compared with the Mann-Whitney test for continuous variables and with the χ2 or Fisher exact test for categorical variables. Kaplan-Meier curves with the log-rank test were used to determine the association of recurrent stroke with NLR and smoking status. The predictive value of NLR in patients with nonacute AICAO was assessed via receiver operating characteristic (ROC) analysis, and the optimal cutoff value was determined.
To identify predictors of recurrent stroke, a Cox regression model was employed both before and after adjustment for potential confounders, including age, hypertension, coronary artery disease, diabetes mellitus, and hypercholesterolemia. All P values were considered significant when <0.05. Statistical analyses were performed with SPSS software version 20.0 (IBM Corp., Armonk, NY, USA).
Results
Participants and baseline characteristics
Of the 281 patients with nonacute internal carotid artery occlusion, 31 were excluded for asymptomatic occlusion, 32 for contralateral internal carotid severe stenosis, 6 for concurrent atrial fibrillation, 2 for internal carotid artery dissection, 3 for refusing any treatment, 45 for successful endovascular recanalization, and 5 for an mRS score >3. Additionally, 21 (11.8%) patients were lost to follow-up and excluded from further analysis. Ultimately, we ultimately included a total of 136 patients with symptomatic nonacute AICAO for analysis (Figure 1).
Clinical outcomes
Over a median follow-up of 12.3 months (IQR, 8.6–21.1 months), ipsilateral recurrent stroke occurred in 17 (12.5%) patients (Figure 2). The 1- and 2-year ipsilateral stroke recurrence rates in the cohort were 8.8% (12/136) and 12.5% (17/136), respectively. All recurrent strokes were ischemic, and no hemorrhagic stroke events were observed. Additionally, 2 patients died from nonvascular events.

The NLR was significantly higher in patients with recurrent stroke (median 3.38, IQR, 2.20–4.95) than in those without it (median 2.39, IQR, 1.82–3.02) (P=0.007). The lymphocyte count was significantly lower in patients with recurrent stroke [median (IQR) ×109/L: 1.55 (1.1–1.95) vs. 1.81 (1.45–2.23); P=0.038]. There was a higher neutrophil count in patients with recurrent stroke [median (IQR) ×109/L: 5.14 [3.58–7.78) vs. 4.54 (3.47–5.31); P=0.076], but not significantly so. Univariate analysis revealed that smoking (P=0.04) and NLR (P=0.01) were also associated with recurrent stroke.
Age, sex, body mass index, hypertension, coronary artery disease, diabetes mellitus, hypercholesterolemia, qualifying events, failed endovascular revascularization, vascular risk factor control status, and neutrophil count were not associated with stroke recurrence (Table 1).
Table 1
| Characteristic | Overall (n=136) | Nonrecurrence (n=119) | Recurrence (n=17) | P value |
|---|---|---|---|---|
| Age (years) | 59 [52–66] | 60 [52–66] | 58 [49–52] | 0.273 |
| Female sex | 20 (14.7) | 17 (14.3) | 3 (17.6) | 0.717 |
| BMI (kg/m2) | 25.1 [23.5–27.1] | 25.0 [23.5–27.7] | 25.2 [23.4–25.9] | 0.934 |
| Hypertension | 86 (63.2) | 74 (62.2) | 12 (70.6) | 0.501 |
| Coronary artery disease | 15 (11.0) | 13 (10.9) | 2 (11.8) | >0.999 |
| Diabetes mellitus | 43 (31.6) | 36 (30.3) | 7 (41.2) | 0.365 |
| Hypercholesterolemia | 11 (8.1) | 10 (8.4) | 1 (5.9) | >0.999 |
| Smoking | 71 (52.2) | 58 (48.7) | 13 (76.5) | 0.032 |
| Qualified events | 0.070 | |||
| TIA | 34 (25.0) | 33 (27.7) | 1 (5.9) | |
| Ischemic stroke | 102 (75.0) | 86 (72.3) | 16 (94.1) | |
| Unsuccessful endovascular revascularization | 0.193 | |||
| No | 109 (80.1) | 93 (78.2) | 16 (94.1) | |
| Yes | 27 (19.9) | 26 (21.8) | 1 (5.9) | |
| Risk factors controlled | 102 (75.0) | 89 (74.8) | 13 (76.5) | >0.999 |
| Neutrophil count (×109/L) | 4.54 [3.47–5.35] | 4.54 [3.50–5.21] | 4.88 [3.69–6.93] | 0.164 |
| Lymphocyte count (×109/L) | 1.79 [1.41–2.21] | 1.81 [1.45–2.23] | 1.55 [1.10–1.95] | 0.038 |
| NLR | 2.45 [1.86–3.26] | 2.39 [1.82–3.02] | 3.38 [2.20–4.95] | 0.007 |
Data are presented as n (%) or median [IQR]. BMI, body mass index; IQR, interquartile range; NLR, neutrophil-lymphocyte ratio; TIA, transient ischemic attack.
Optimal NLR cutoff value
We constructed ROC curves to evaluate the optimal cutoff and prediction value of NLR and lymphocyte count for stroke recurrence. The ROC curve analysis identified an optimal NLR cutoff of 3.36, with a specificity of 0.815 and sensitivity of 0.588. The area under the curve (AUC) for stroke recurrence was 0.67 [95% confidence interval (CI): 0.55–0.80; P=0.01; Figure 3]. This value indicates a modest discriminatory ability of the NLR in predicting stroke recurrence in patients with symptomatic nonacute AICAO. Based on the NLR value, patients were divided into two groups, with 22.8% (n=31) in the NLR >3.36 group and 77.2% (n=105) in the NLR ≤3.36 group. Notably, 52.9% (9/17) of the patients with recurrent stroke had an NLR >3.36, whereas only 18.5% (22/119) of the patients with nonrecurrent stroke had an NLR >3.36.

Predictors of stroke recurrence
The 1-year stroke recurrence rates were 22.5% (7/31) and 5.7% (6/105) in the NLR >3.36 group and the NLR ≤3.36 group, respectively (Table S1). We evaluated the comparison of stroke recurrence between the NLR >3.36 and NLR ≤3.36 groups using Kaplan-Meier curves and the log-rank test. The cumulative incidence of recurrent stroke in the NLR >3.36 group was statistically higher than that in the NLR ≤3.36 group (P=0.039), with the incidence difference between the two groups increasing over time (Figure 4).

In the univariate Cox proportional hazards analyses, smoking [hazard ratio (HR) 3.06, 95% CI: 1.00–9.41] and NLR >3.36 (HR 3.83, 95% CI: 1.48–9.94) were associated with stroke recurrence. After adjustments were made for age and smoking, NLR >3.36 remained a significant marker for stroke recurrence (HR 4.17, 95% CI: 1.59–10.91; Table 2). With 17 events and an observed of HR ≈4.2, the study had ≈70% power at α=0.05; thus, negative confounders could have been missed. Even after confounding variables were accounted for, the NLR, considered to be a continuous variable, remained significantly correlated with stroke recurrence of (HR 1.99, 95% CI: 1.49–2.66; Table 2).
Table 2
| Characteristic | Univariate | Multivariate | |||
|---|---|---|---|---|---|
| HR | P value | Adjusted HR | P value | ||
| Age (years) | 0.98 | 0.41 | 0.98 | – | |
| Sex | 0.93 | 0.91 | – | – | |
| BMI | 0.97 | 0.81 | – | – | |
| Hypertension | 1.4 | 0.53 | – | – | |
| Coronary artery disease | 1.08 | 0.92 | – | – | |
| Diabetes mellitus | 1.49 | 0.42 | – | – | |
| Hypercholesterolemia | 0.95 | 0.96 | – | – | |
| Smoking | 3.06 | 0.05 | 3.25 | 0.04 | |
| NLR >3.36 | 3.83 | 0.01 | 4.17 | 0.01 | |
| NLR | 1.83 | 0.01 | 1.83 | 0.01 | |
| Endovascular treatment | 0.26 | 0.19 | – | – | |
Multivariable Cox model constructed with adjustments for the factors of age, smoking, and NLR >3.36. BMI, body mass index; HR, hazard ratio; NLR, neutrophil-lymphocyte ratio.
Discussion
In this study, NLR was found to be an independent predictor of stroke recurrence in patients with symptomatic nonacute AICAO. The optimal cutoff value of NLR in predicting stroke recurrence was determined to be 3.36. Patients with NLR >3.36 were associated with an approximately threefold risk of stroke recurrence. However, the optimal NLR cutoff value (3.36) was derived from the same data set used for statistical testing. Consequently, the threshold may be data-specific and could vary in other populations. External validation in an independent, multicenter cohort is needed before this cutoff can be adopted clinically.
The primary treatment of symptomatic nonacute AICAO is aggressive medical treatment. Randomized control trials have failed to show the superiority of surgical treatment over medical treatment even in highly selected patients and surgical centers (3,4). Aside from high perioperative events, the better-than-expected outcome of medical therapy nullifies the potential benefit of surgery (4). However, there may be subgroups of patients not well represented in these clinical trials, in whom the recurrence rate is high despite optimal medical treatment (19). Revascularization may have a potential role in reducing the recurrence rate. Therefore, identifying the predictors of stroke recurrence is crucial for both clinical management and the design of future clinical trials. Our results suggest that NLR is an independent predictor of stroke recurrence. Patients with NLR >3.36 had a significantly higher recurrence rate than did patients with NLR <3.36 (1-year stroke recurrence rate: 22.5% vs. 5.7%).
Previous research has indicated that age and cerebral hemodynamic compromise are predictors of stroke recurrence (5,20,21). In our study, the hemodynamic status was not evaluated and not used as a patient selection criterion. It is important to note that the high-risk group in our study was defined by inflammation, as indicated by NLR, rather than by hemodynamics. Therefore, a direct comparison of recurrence rates with trials that exclusively enrolled patients with proven hemodynamic failure, such as the Carotid Occlusion Surgery Study (COSS) and Carotid or Middle Cerebral Arterial Occlusion Surgery Study (CMOSS), would not be valid. These trials focused on patients with hemodynamic compromise, whereas our study identified a different high-risk pathway related to inflammation. Furthermore, we did not evaluate the hemodynamic status and did not use it as a criterion for patient selection. Because both hemodynamic compromise and systemic inflammation (as reflected by NLR) may independently or synergistically increase the risk of stroke recurrence, we cannot exclude the possibility that part of the observed association is mediated by covert hemodynamic failure. Future studies should therefore combine perfusion imaging [computed tomographic perfusion (CTP), magnetic resonance (MR) perfusion, or positron emission tomography (PET)] with inflammatory biomarkers to clarify the interaction between these two pathways. Our findings indicate that the efficacy of current medical therapy is suboptimal in patients with high NLR. Future studies are required to investigate the effect of revascularization in these high-risk patients.
Emerging evidence suggests that systemic inflammatory markers, including NLR, systemic immune-inflammation index (SII), systemic inflammation response index (SIRI), and lymphocyte-monocyte ratio (LMR), are closely associated with the severity, progression, and short-term prognosis of patients with ischemic stroke (22-25). As an easily accessible indicator of chronic low-level inflammation (26), the NLR has the potential to act as a bridge between the diagnosis of chronic low-level inflammation and that of atherosclerosis. In patients with atherosclerosis, lymphocyte counts may decrease in response to corticosteroids that are induced by stressful conditions (27). Lymphocytopenia may also be a result of a large consumption of lymphocytes due to the frequent infiltration of lymphocytes into plaques during the development of atherosclerosis (28). It was suggested that by secreting anti-inflammatory cytokines, some lymphocyte subtypes, such as type 2 helper T cells, promote the atherosclerotic plaque healing process (29). Therefore, a decreased lymphocyte count caused by chronic low-level inflammation might accelerate atherosclerosis plaque progression. Meanwhile, neutrophils and lymphocytes may have different roles in the pathophysiologic development of atherosclerosis. Neutrophils could accumulate on the vascular wall and cause vascular chronic low-level inflammation, which may lead to endothelial dysfunction and subsequent atherosclerosis (30,31). Studies have reported that NLR is associated with large-artery (both extracranial and intracranial) atherosclerosis (8,16). An analysis of five randomized control trials demonstrated that the baseline NLR can predict the risk of major adverse cardiovascular events, with the risk increasing by 9% to 31% per-quartile increment in NLR (9). Although experimental data show that neutrophil-derived myeloperoxidase, reactive oxygen species, and neutrophils release extracellular traps can accelerate endothelial dysfunction and plaque necrosis, they only suggest—and do not prove—a causal role (32). Meanwhile, a large-scale Mendelian randomization study found no significant effect of genetically predicted NLR on carotid intima–media thickness or coronary disease risk, suggesting that NLR is primarily a marker of systemic inflammatory burden rather than a direct causal driver (33).
Xue et al. reported that the NLR is associated with stroke recurrence and poor prognosis (11). Other research indicated that the NLR is associated with recurrent ischemic stroke in patients with embolic stroke of undetermined source (10). In patients with symptomatic nonacute AICAO, a potential mechanism for stroke recurrence is low-level inflammation (which could be indicated by NLR) accelerating intracranial atherosclerosis, which in turn impairs the compensatory pathways that redistribute flow and impair the protection of brain tissue at risk from ischemic stroke (34).
An AUC of 0.703 denotes modest discriminative performance. To achieve clinically useful prediction, we envision integrating NLR with existing tools including (i) perfusion-/collateral-imaging metrics (CTP, MR-perfusion, and PET) to capture hemodynamic risk and (ii) validated risk scores (e.g., Essen, Stroke Prognosis Instrument II, or COSS-based clinical hyperperfusion score) that already contain demographic and vascular variables. Preliminary evidence from studies on large-artery stroke indicates that adding systemic inflammation markers to imaging or risk score models can increase the AUC by ≈0.08–0.12 and improve reclassification (category-free net reclassification improvement 0.25–0.40). Future prospective studies should therefore construct multimodal panels (imaging + clinical score + NLR) and quantify the incremental value with net reclassification improvement and integrated discrimination improvement.
Of the 157 patients enrolled after initial exclusions, 21 (13.4%) patients were lost to follow-up. This rate of attrition is substantial and might have introduced bias into our findings. Patients who are lost to follow-up are often systematically different from those who remain in the study. They may have more severe illness, be less compliant with treatment, or be healthier and have moved away. These differences could potentially affect the generalizability and validity of our results. To mitigate this concern, we conducted a detailed comparison of the baseline characteristics of the 21 patients who were lost to follow-up and the 136 patients who remained in the study. The results of this comparison are summarized in Table S2. We found no significant differences in age, sex, hypertension, diabetes mellitus, hyperlipidemia, or baseline NLR between the two groups. This suggests that the patients who were lost to follow-up were not systematically different from those who remained, reducing the likelihood of significant bias. Although the baseline characteristics did not differ between the lost and retained participants, unmeasured differences (e.g., socioeconomic status, adherence, or occurrent vascular events) could have still biased our estimates. Future prospective studies should employ centralized registries or telephone tracing to minimize loss to follow-up and allow credible sensitivity analyses.
This study involved several limitations that should be acknowledged. First, there were potential sources of bias. The retrospective, single-center design inherently precludes causal inference. Second, the low number of outcome events (only 17 recurrent strokes) restricted the complexity of the multivariate analysis. This relatively small number of events may reduce the statistical power and the ability to detect significant associations. Future studies with larger sample sizes and more detailed data collection are needed to address these limitations and further validate our findings. Third, the lack of validation for the NLR threshold is a significant limitation of our study. Although our initial findings are promising, they need to be confirmed through additional validation efforts.
Conclusions
Our study found that in patients with symptomatic nonacute AICAO, an NLR >3.36 was a predictor for recurrent stroke after aggressive medical treatment. This simple, inexpensive, and immediately obtainable inflammatory marker can help identify patients at high risk for stroke recurrence.
Acknowledgments
None.
Footnote
Reporting Checklist: The authors have completed the STROBE reporting checklist. Available at https://qims.amegroups.com/article/view/10.21037/qims-2025-1437/rc
Data Sharing Statement: Available at https://qims.amegroups.com/article/view/10.21037/qims-2025-1437/dss
Funding: This work was supported by
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://qims.amegroups.com/article/view/10.21037/qims-2025-1437/coif). N.M. reports support from the National Natural Science Foundation of China (grant No. 82171894). W.F. reports support from the National Natural Science Foundation of China (grant No. 82401517). The other authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The study was conducted in accordance with the Declaration of Helsinki and its subsequent amendments. The study was approved by the Institutional Review Board of Beijing Tiantan Hospital (No. KY2022-077-02) and informed consent was obtained from all individual participants.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Powers WJ. Atherosclerotic Carotid Artery Occlusion. Curr Treat Options Neurol 2003;5:381-9. [Crossref] [PubMed]
- Paciaroni M, Caso V, Venti M, Milia P, Kappelle LJ, Silvestrelli G, Palmerini F, Acciarresi M, Sebastianelli M, Agnelli G. Outcome in patients with stroke associated with internal carotid artery occlusion. Cerebrovasc Dis 2005;20:108-13. [Crossref] [PubMed]
- Ma Y, Wang T, Wang H, Amin-Hanjani S, Tong X, Wang J, et al. Extracranial-Intracranial Bypass and Risk of Stroke and Death in Patients With Symptomatic Artery Occlusion: The CMOSS Randomized Clinical Trial. JAMA 2023;330:704-14. [Crossref] [PubMed]
- Powers WJ, Clarke WR, Grubb RL Jr, Videen TO, Adams HP Jr, Derdeyn CP. Extracranial-intracranial bypass surgery for stroke prevention in hemodynamic cerebral ischemia: the Carotid Occlusion Surgery Study randomized trial. JAMA 2011;306:1983-92. [Crossref] [PubMed]
- Yuan HW, Ji RJ, Wang AG, Lin YJ, Chen HF, Xu ZQ, Peng ZY, Luo BY. Age and composite end-point events in medium follow-up of patients with carotid artery total occlusion using drug therapy. Atherosclerosis 2017;265:184-9. [Crossref] [PubMed]
- Ross R. Atherosclerosis--an inflammatory disease. N Engl J Med 1999;340:115-26. [Crossref] [PubMed]
- Song M, Graubard BI, Rabkin CS, Engels EA. Neutrophil-to-lymphocyte ratio and mortality in the United States general population. Sci Rep 2021;11:464. [Crossref] [PubMed]
- Chung D, Lee KO, Choi JW, Kim NK, Kim OJ, Kim SH, Oh SH, Kim WC. Blood Neutrophil/Lymphocyte Ratio Is Associated With Cerebral Large-Artery Atherosclerosis but Not With Cerebral Small-Vessel Disease. Front Neurol 2020;11:1022. [Crossref] [PubMed]
- Adamstein NH, MacFadyen JG, Rose LM, Glynn RJ, Dey AK, Libby P, Tabas IA, Mehta NN, Ridker PM. The neutrophil-lymphocyte ratio and incident atherosclerotic events: analyses from five contemporary randomized trials. Eur Heart J 2021;42:896-903. [Crossref] [PubMed]
- Li TYW, Sia CH, Chan BPL, Ho JSY, Leow AS, Chan MY, Kojodjojo P, Galupo MJ, Teoh HL, Sharma VK, Seet RCS, Yeo LLL, Tan BY. Neutrophil-Lymphocyte and Platelet-Lymphocyte Ratios Are Associated with Recurrent Ischemic Stroke in Patients with Embolic Stroke of Undetermined Source. J Stroke 2022;24:421-4. [Crossref] [PubMed]
- Xue J, Huang W, Chen X, Li Q, Cai Z, Yu T, Shao B. Neutrophil-to-Lymphocyte Ratio Is a Prognostic Marker in Acute Ischemic Stroke. J Stroke Cerebrovasc Dis 2017;26:650-7. [Crossref] [PubMed]
- Celikbilek A, Ismailogullari S, Zararsiz G. Neutrophil to lymphocyte ratio predicts poor prognosis in ischemic cerebrovascular disease. J Clin Lab Anal 2014;28:27-31. [Crossref] [PubMed]
- Gökhan S, Ozhasenekler A, Mansur Durgun H, Akil E, Ustündag M, Orak M. Neutrophil lymphocyte ratios in stroke subtypes and transient ischemic attack. Eur Rev Med Pharmacol Sci 2013;17:653-7.
- Tokgoz S, Kayrak M, Akpinar Z, Seyithanoğlu A, Güney F, Yürüten B. Neutrophil lymphocyte ratio as a predictor of stroke. J Stroke Cerebrovasc Dis 2013;22:1169-74. [Crossref] [PubMed]
- Broderick JP, Adeoye O, Elm J. Evolution of the Modified Rankin Scale and Its Use in Future Stroke Trials. Stroke 2017;48:2007-12. [Crossref] [PubMed]
- Arbel Y, Finkelstein A, Halkin A, Birati EY, Revivo M, Zuzut M, Shevach A, Berliner S, Herz I, Keren G, Banai S. Neutrophil/lymphocyte ratio is related to the severity of coronary artery disease and clinical outcome in patients undergoing angiography. Atherosclerosis 2012;225:456-60. [Crossref] [PubMed]
- Kleindorfer DO, Towfighi A, Chaturvedi S, Cockroft KM, Gutierrez J, Lombardi-Hill D, Kamel H, Kernan WN, Kittner SJ, Leira EC, Lennon O, Meschia JF, Nguyen TN, Pollak PM, Santangeli P, Sharrief AZ, Smith SC Jr, Turan TN, Williams LS. 2021 Guideline for the Prevention of Stroke in Patients With Stroke and Transient Ischemic Attack: A Guideline From the American Heart Association/American Stroke Association. Stroke 2021;52:e364-467. [Crossref] [PubMed]
- Sacco RL, Kasner SE, Broderick JP, Caplan LR, Connors JJ, Culebras A, et al. An updated definition of stroke for the 21st century: a statement for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 2013;44:2064-89. [Crossref] [PubMed]
- Reynolds MR, Derdeyn CP, Grubb RL Jr, Powers WJ, Zipfel GJ. Extracranial-intracranial bypass for ischemic cerebrovascular disease: what have we learned from the Carotid Occlusion Surgery Study? Neurosurg Focus 2014;36:E9. [Crossref] [PubMed]
- Klijn CJ, Kappelle LJ, Tulleken CA, van Gijn J. Symptomatic carotid artery occlusion. A reappraisal of hemodynamic factors. Stroke 1997;28:2084-93. [Crossref] [PubMed]
- Grubb RL Jr, Derdeyn CP, Fritsch SM, Carpenter DA, Yundt KD, Videen TO, Spitznagel EL, Powers WJ. Importance of hemodynamic factors in the prognosis of symptomatic carotid occlusion. JAMA 1998;280:1055-60. [Crossref] [PubMed]
- Jiang L, Cai X, Yao D, Jing J, Mei L, Yang Y, Li S, Jin A, Meng X, Li H, Wei T, Wang Y, Pan Y, Wang Y. Association of inflammatory markers with cerebral small vessel disease in community-based population. J Neuroinflammation 2022;19:106. [Crossref] [PubMed]
- Huang C, Geng J, Fan J, Tian B, Wang K, Zhang Y, Zhou X, Zhu X, Sun Z. Atherogenic and Inflammatory Markers in Recent Small Subcortical Infarcts: Associations with Location, Morphology, and Short-Term Recurrence. J Inflamm Res 2025;18:5881-93. [Crossref] [PubMed]
- Nordestgaard AT, Moorthy MV, Cook NR, Rifai N, Lee IM, Buring JE, Ridker PM. High-sensitivity C-reactive protein, LDL cholesterol, lipoprotein(a) and 30-year risk of stroke in healthy women: a prospective, longitudinal cohort study. Lancet Neurol 2025;24:920-30. [Crossref] [PubMed]
- Wang Y, Zhang Z, Hang X, Wang W. Associations of Inflammatory Markers With Neurological Dysfunction and Prognosis in Patients With Progressive Stroke. Eur J Neurol 2025;32:e70080. [Crossref] [PubMed]
- Balta S, Kurtoglu E, Kucuk U, Demirkol S, Ozturk C. Neutrophil-lymphocyte ratio as an important assessment tool. Expert Rev Cardiovasc Ther 2014;12:537-8. [Crossref] [PubMed]
- Tamhane UU, Aneja S, Montgomery D, Rogers EK, Eagle KA, Gurm HS. Association between admission neutrophil to lymphocyte ratio and outcomes in patients with acute coronary syndrome. Am J Cardiol 2008;102:653-7. [Crossref] [PubMed]
- Glaudemans AW, Bonanno E, Galli F, Zeebregts CJ, de Vries EF, Koole M, Luurtsema G, Boersma HH, Taurino M, Slart RH, Signore A. In vivo and in vitro evidence that 99mTc-HYNIC-interleukin-2 is able to detect T lymphocytes in vulnerable atherosclerotic plaques of the carotid artery. Eur J Nucl Med Mol Imaging 2014;41:1710-9. [Crossref] [PubMed]
- Vergallo R, Crea F. Atherosclerotic Plaque Healing. N Engl J Med 2020;383:846-57. [Crossref] [PubMed]
- Meng LB, Yu ZM, Guo P, Wang QQ, Qi RM, Shan MJ, Lv J, Gong T. Neutrophils and neutrophil-lymphocyte ratio: Inflammatory markers associated with intimal-media thickness of atherosclerosis. Thromb Res 2018;170:45-52. [Crossref] [PubMed]
- Qi H, Yang S, Zhang L. Neutrophil Extracellular Traps and Endothelial Dysfunction in Atherosclerosis and Thrombosis. Front Immunol 2017;8:928. [Crossref] [PubMed]
- Yalcinkaya M, Fotakis P, Liu W, Endo-Umeda K, Dou H, Abramowicz S, Xiao T, Libby P, Wang N, Tall AR, Westerterp M. Cholesterol accumulation in macrophages drives NETosis in atherosclerotic plaques via IL-1β secretion. Cardiovasc Res 2023;119:969-81. [Crossref] [PubMed]
- Lai B, Huang B, Li L. Causal relationship between inflammatory markers and left ventricle geometry and function: A 2-sample Mendelian randomization study. Medicine (Baltimore) 2024;103:e38735. [Crossref] [PubMed]
- Yeo LL, Kong WY, Paliwal P, Teoh HL, Seet RC, Soon D, Rathakrishnan R, Ong V, Lee TH, Wong HF, Chan BP, Leow WK, Yuan C, Ting E, Gopinathan A, Tan BY, Sharma VK. Intravenous Thrombolysis for Acute Ischemic Stroke due to Cervical Internal Carotid Artery Occlusion. J Stroke Cerebrovasc Dis 2016;25:2423-9. [Crossref] [PubMed]