
Imaging characteristics of resting myocardial perfusion imaging and myocardial glucose metabolic imaging in stress cardiomyopathy
Introduction
Stress cardiomyopathy (SCM), first described by Sato in 1991, is a syndrome of temporary ventricular insufficiency with clinical manifestations similar to those of acute myocardial infarction (AMI) (1). SCM is also known as Takotsubo syndrome due to the resemblance of the left ventricular angiographic manifestations to Japanese octopus traps and is prevalent in menopausal women (2). The etiology of SCM has not been fully clarified. The majority of patients with SCM have a history of stress, including mental stress and physical stress, while some have no clear stressful events (3), with mental stress predominating in women and physical stress in men (4). Normal coronary angiography and regional wall dysfunction with bulging on left ventriculography are regarded as the standard criteria for excluding or confirming SCM (5).
Single-photon emission computed tomography (SPECT) myocardial perfusion imaging (MPI) is a widely used and noninvasive method for detecting myocardial ischemia and myocardial viability (6,7). Gated MPI can simultaneously provide myocardial perfusion and left ventricular functional indicators such as wall motion and wall thickening (8,9). However, few studies on the MPI characteristics of SCM have been conducted, and there is controversy regarding whether myocardial perfusion is reduced in areas with abnormal wall function (10-13). In addition, myocardial glucose metabolic imaging can reflect myocardial energy metabolism and can be used to examine the pathophysiological mechanism of SCM (14). Our center recently admitted a patient with confirmed SCM and complete clinical data who underwent MPI and myocardial glucose metabolic imaging. The relevant image characteristics have been summarized in this report.
Case presentation
All procedures performed in this study were in accordance with the ethical standards of the institutional and/or national research committee(s) and with the Declaration of Helsinki and its subsequent amendments. Written informed consent was obtained from the patient for publication of this article and accompanying images. A copy of the written consent is available for review by the editorial office of this journal.
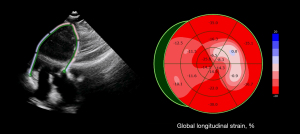
The patient was a 61-year-old female who presented with chest pain, accompanied by radiating pain in the left back, after an emotional event and then immediately arrived at the Third Affiliated Hospital of Soochow University. The patient had no history of hypertension, diabetes, or hyperlipidemia. Electrocardiography (ECG) showed sinus bradycardia, aberrant ventricular conduction, QTc prolongation, q wave (leads I and aVL), atrial premature beats, and T-wave inversion (leads I–II, aVR, aVL, and V3–V6). At the time of admission, the level of high-sensitivity cardiac troponin I (hs-cTnI) was 5.175 ng/mL (normal value 0–0.04 ng/mL) and that of N-terminal pro B-type natriuretic peptide (NT-proBNP) was 4,860 ng/L (normal value 300–450 ng/L). Six hours later, the level of hs-cTnI was 3.971 ng/mL. Left ventricular speckle tracking echocardiography and global longitudinal strain analysis show reduced wall motion in the anterior wall near the apex and the lateral wall of the left ventricle, as shown in Figure 1; the left ventricular ejection fraction (LVEF) was 48%. The four-chamber images at the end of systole and diastole are presented in Figure S1.
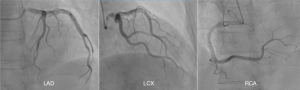
Emergency coronary angiography was performed, which showed no significant stenosis in the left main coronary artery, left anterior descending coronary artery, left circumflex coronary artery, or right coronary artery, as shown in Figure 2. The patient’s InterTAK diagnostic score was 78, and based on the medical history, clinical presentation, laboratory results, ECG, and angiographic findings, a diagnosis of SCM was made.
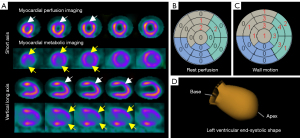
One day later, the patient underwent 1-day rest-state SPECT MPI combined with 18F-fluorodeoxyglucose (FDG) positron emission tomography (PET) myocardial metabolic imaging in our department to evaluate myocardial perfusion and glucose metabolism. Resting-stated 99mtechnetium-sestamibi (99mTc-MIBI)-gated SPECT MPI was performed first. PET myocardial metabolic imaging was performed via injection of 18F-FDG directly into the patient without fasting measures, and imaging was performed 1 hour after injection. Resting-state MPI showed mildly decreased myocardial perfusion in the middle segment of the anterior wall and the anterior wall near the apex. Myocardial metabolic imaging showed decreased myocardial glucose metabolism in the middle segment of the anterior wall, the anterior wall near the apex, the apex, and the inferior wall near the apex as compared to the basal segment of the inferior wall and the septal wall. Myocardial blood perfusion and glucose metabolism were both reduced, exhibiting characteristics of perfusion-metabolism match. The images from the abovementioned scans are provided in Figure 3A. Semiquantitative analysis was applied to the resting-state MPI, and quantitative perfusion SPECT (QPS) software analysis showed that three myocardial segments had abnormal perfusion, with a total score of 5 (Figure 3B). Meanwhile, quantitative gated SPECT (QGS) software analysis showed abnormal wall motion in 11 myocardial segments. The summed wall motion abnormality score was 23 (Figure 3C). The range and degree of wall motion dysfunction were significantly greater than those of myocardial perfusion, indicating perfusion-function mismatch. The end-systolic shape based on rest MPI revealed typical ballooning of the apical segments (Figure 3D).
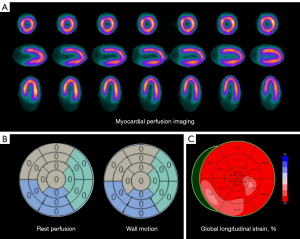
During hospitalization, symptomatic treatment including coronary dilation, anticoagulation, and medications to improve myocardial metabolism were administered. After 3 days, the patient’s chest pain symptoms improved. Three months after discharge, echocardiography and MPI were performed to assess recovery. Resting-state MPI showed that the decreased perfusion area in the middle segment of the anterior wall and the anterior wall near the apex had returned to normal (Figure 4A). QPS and QGS semiquantitative software analysis indicated normal perfusion and wall motion (Figure 4B). The echocardiography and global longitudinal strain analysis showed that the areas of reduced wall motion in the anterior wall near the apex and the lateral wall had recovered (Figure 4C), with the LVEF being 61%.
Discussion
This case illustrates the MPI and myocardial metabolic imaging characteristics of SCM. In our patient, myocardial perfusion and glucose metabolism were decreased, which was accompanied by abnormal wall function. In addition, the range and extent of wall motion were significantly greater than those of myocardial perfusion.
According to the location of abnormal wall motion, SCM can be divided into the apical type, middle ventricular type, basal type, and localized type (15), with the apical type being the most common. The pathophysiology of SCM has not been fully elucidated, but the bulk of evidence indicates that sympathetic nerve stimulation is central to its pathogenesis (16). Other possible mechanisms include reversible coronary microcirculation dysfunction, epicardial coronary spasm, and the toxic effects of excessive catecholamine on myocardial cells. Catecholamines and endothelin mainly exert their vasoconstrictor effects in the coronary microvasculature, where α1-receptors and endothelin receptor type A are predominant. This indicates that acute microcirculatory dysfunction might play a central role in SCM. Moreover, in the acute stage of SCM, there is a decrease in the level of microRNA 125a-5p. Concurrently, the plasma levels of its target, endothelin-1, are elevated, which is consistent with the hypothesis of microvascular spasms (17). During the acute phase of SCM, similar to the reduction in coronary flow reserve, microvascular blood flow may also decline. SCM often needs to be distinguished from vasospastic angina (VSA), which is clinically characterized by recurrent episodes of resting-state chest pain, often occurring at the same time each day and can be relieved by nitroglycerin (18). VSA is more indicated when typical angina symptoms, transient ST segment elevation on ECG, and normal coronary arteries on angiography are present (19). In contrast, SCM is typically triggered by emotional or physical stress and manifests as acute chest pain, shortness of breath, and ECG changes similar to those of AMI, apical balloons on echocardiography, and the presence of elevated cardiac biomarkers.
Resting-state MPI was performed in this patient to assess myocardial injury and revealed a mild reduction in myocardial perfusion in the middle segment of the anterior wall and the anterior wall near the apex. However, the QGS software analysis showed abnormal wall motion in 11 myocardial segments. The range and degree of wall motion are significantly greater than those of myocardial perfusion. This mild decrease in perfusion indicated that the extent of myocardial injury was small, there was no severe transmural myocardial infarction, and the apical myocardium was still alive, which was a critical basis for the patient’s recovery of wall motion and myocardial function. Echocardiography can assist in the diagnosis of SCM. Typical echocardiographic findings of SCM include apical ballooning, myocardial hypokinesia, or dysfunction in the middle segment of the left ventricle (20). In our case, we observed abnormal segments of wall motion using left ventricular speckle tracking echocardiography and longitudinal strain analysis techniques, and the associated indicators appeared to recover during subsequent follow-up.
Myocardial metabolic imaging in this patient showed decreased myocardial glucose metabolism in the middle segment of the anterior wall, anterior wall near the apex, apex, and inferior wall near the apex. Perfusion-metabolism match is often used in patients with ischemic heart disease to describe the scar myocardium. Here, we use it to express the imaging characteristics of both reduced perfusion and metabolism. Studies have shown that reduced myocardial glucose metabolism in abnormal wall function segments in patients with SCM is to some extent related to a catecholamine surge after stress (21). In addition, the density of β-adrenergic receptors is greater in the apex than in the base, resulting in a more intense response to transient catecholamines in the apex (22). This uneven distribution of β-receptors may explain the association between a transient catecholamine surge and decreased glucose metabolism in the apex of individuals with SCM, but the specific mechanisms underlying this remain to be investigated.
Certain limitations to this study should be noted. This was only a review of a single patient, and a large-sample study is required to validate our findings. In addition, no further analysis was conducted on the molecular pathological mechanisms of reduced myocardial perfusion and glucose metabolism.
Conclusions
Mildly reduced myocardial perfusion and reduced glucose metabolism in segments with abnormal wall function may be imaging features characteristic to SCM. MPI and myocardial metabolic imaging may help the diagnosis and study of the pathophysiological mechanisms in SCM.
Acknowledgments
None.
Footnote
Funding: This work was supported by
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://qims.amegroups.com/article/view/10.21037/qims-2025-413/coif). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. All procedures performed in this study were in accordance with the ethical standards of the institutional and/or national research committee(s) and with the Declaration of Helsinki and its subsequent amendments. Written informed consent was obtained from the patient for publication of this article and accompanying images. A copy of the written consent is available for review by the editorial office of this journal.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Kurisu S, Inoue I, Kawagoe T, et al. Myocardial perfusion and fatty acid metabolism in patients with tako-tsubo-like left ventricular dysfunction. J Am Coll Cardiol 2003;41:743-8. [Crossref] [PubMed]
- Kato K, Lyon AR, Ghadri JR, Templin C. Takotsubo syndrome: aetiology, presentation and treatment. Heart 2017;103:1461-9. [Crossref] [PubMed]
- Isogai T, Yasunaga H, Matsui H, Tanaka H, Ueda T, Horiguchi H, Fushimi K. Out-of-hospital versus in-hospital Takotsubo cardiomyopathy: analysis of 3719 patients in the Diagnosis Procedure Combination database in Japan. Int J Cardiol 2014;176:413-7. [Crossref] [PubMed]
- Kurisu S, Inoue I, Kawagoe T, Ishihara M, Shimatani Y, Nakama Y, Kagawa E, Dai K, Ikenaga H. Presentation of Tako-tsubo cardiomyopathy in men and women. Clin Cardiol 2010;33:42-5. [Crossref] [PubMed]
- Ghadri JR, Wittstein IS, Prasad A, Sharkey S, Dote K, Akashi YJ, et al. International Expert Consensus Document on Takotsubo Syndrome (Part I): Clinical Characteristics, Diagnostic Criteria, and Pathophysiology. Eur Heart J 2018;39:2032-46. [Crossref] [PubMed]
- Wang F, Yuan H, Lv J, Han X, Zhou Z, Lu W, Lu L, Jiang L. Stress-only versus rest-stress SPECT MPI in the detection and diagnosis of myocardial ischemia and infarction by machine learning. Nucl Med Commun 2024;45:35-44. [Crossref] [PubMed]
- Padrón K, Peix A, Cabrera L, Garcia J, Rodriguez L, Carrillo R, Mena E, Fernandez Y. Could myocardial viability be related to left ventricular dyssynchrony? Simultaneous evaluation by gated SPECT-MPI. J Nucl Cardiol 2020;27:1158-67. [Crossref] [PubMed]
- Zhang F, Wang J, Shao X, Yang M, Qian Y, Yang X, Wu Z, Li S, Xin W, Shi Y, Liu B, Yu W, He Z, Zhou W, Wang Y. Incremental value of myocardial wall motion and thickening to perfusion alone by gated SPECT myocardial perfusion imaging for viability assessment in patients with ischemic heart failure. J Nucl Cardiol 2021;28:2545-56. [Crossref] [PubMed]
- Nkoulou R, Wolfrum M, Pazhenkottil AP, Fiechter M, Buechel RR, Gaemperli O, Kaufmann PA. Gated SPECT myocardial perfusion imaging with cadmium-zinc-telluride detectors allows real-time assessment of dobutamine-stress-induced wall motion abnormalities. J Nucl Cardiol 2019;26:1734-42. [Crossref] [PubMed]
- Cimarelli S, Imperiale A, Ben-Sellem D, Rischner J, Detour J, Morel O, Ohlmann P, Constantinesco A. Nuclear medicine imaging of takotsubo cardiomyopathy: typical form and midventricular ballooning syndrome. J Nucl Cardiol 2008;15:137-41. [Crossref] [PubMed]
- Cimarelli S, Sauer F, Morel O, Ohlmann P, Constantinesco A, Imperiale A. Transient left ventricular dysfunction syndrome: patho-physiological bases through nuclear medicine imaging. Int J Cardiol 2010;144:212-8. [Crossref] [PubMed]
- Ghadri JR, Dougoud S, Maier W, Kaufmann PA, Gaemperli O, Prasad A, Lüscher TF, Templin C A. PET/CT-follow-up imaging study to differentiate takotsubo cardiomyopathy from acute myocardial infarction. Int J Cardiovasc Imaging 2014;30:207-9. [Crossref] [PubMed]
- Rodríguez LO, Fernández RG, Luis JJ, González AP. Tako-tsubo syndrome: atypical nuclear medicine findings. World J Nucl Med 2012;11:35-8. [Crossref] [PubMed]
- Yoshida T, Hibino T, Kako N, Murai S, Oguri M, Kato K, Yajima K, Ohte N, Yokoi K, Kimura G. A pathophysiologic study of tako-tsubo cardiomyopathy with F-18 fluorodeoxyglucose positron emission tomography. Eur Heart J 2007;28:2598-604. [Crossref] [PubMed]
- Templin C, Ghadri JR, Diekmann J, Napp LC, Bataiosu DR, Jaguszewski M, et al. Clinical Features and Outcomes of Takotsubo (Stress) Cardiomyopathy. N Engl J Med 2015;373:929-38. [Crossref] [PubMed]
- Sharkey SW, Windenburg DC, Lesser JR, Maron MS, Hauser RG, Lesser JN, Haas TS, Hodges JS, Maron BJ. Natural history and expansive clinical profile of stress (tako-tsubo) cardiomyopathy. J Am Coll Cardiol 2010;55:333-41. [Crossref] [PubMed]
- Jaguszewski M, Osipova J, Ghadri JR, Napp LC, Widera C, Franke J, Fijalkowski M, Nowak R, Fijalkowska M, Volkmann I, Katus HA, Wollert KC, Bauersachs J, Erne P, Lüscher TF, Thum T, Templin C. A signature of circulating microRNAs differentiates takotsubo cardiomyopathy from acute myocardial infarction. Eur Heart J 2014;35:999-1006. [Crossref] [PubMed]
- Chahin M, Zoltowska DM, Al-Turk B, Suryadevara S. Vasospastic angina on coronary angiography. BMJ Case Rep 2020;13:e237753. [Crossref] [PubMed]
- Saito Y, Saito Y, Kato K, Kobayashi Y. Gender differences in factors associated with vasospastic angina. Int J Cardiol 2022;349:7-11. [Crossref] [PubMed]
- Ghadri JR, Cammann VL, Napp LC, Jurisic S, Diekmann J, Bataiosu DR, Seifert B, Jaguszewski M, Sarcon A, Neumann CA, Geyer V, Prasad A, Bax JJ, Ruschitzka F, Lüscher TF, Templin C. International Takotsubo (InterTAK) Registry. Differences in the Clinical Profile and Outcomes of Typical and Atypical Takotsubo Syndrome: Data From the International Takotsubo Registry. JAMA Cardiol 2016;1:335-40. [Crossref] [PubMed]
- Egert S, Nguyen N, Schwaiger M. Contribution of alpha-adrenergic and beta-adrenergic stimulation to ischemia-induced glucose transporter (GLUT) 4 and GLUT1 translocation in the isolated perfused rat heart. Circ Res 1999;84:1407-15. [Crossref] [PubMed]
- Huang MT, Lee CF, Dobson GP. Epinephrine enhances glycogen turnover and depresses glucose uptake in vivo in rat heart. FASEB J 1997;11:973-80. [Crossref] [PubMed]


