
Prenatal diagnosis of caudal regression syndrome with maternal diabetes mellitus via ultrasound: a case description
Introduction
Caudal regression syndrome (CRS) is a rare congenital anomaly affecting the neural tube and lower spine. It is also known as sacral agenesis or caudal dysplasia, and it is most frequently linked to genitourinary abnormalities, lower extremity deformities, and neural tube defects (1). In the general population, the incidence of CRS ranges from 1 to 2.5 per 100,000 (2), and 200–250 times more fetal CRS cases occur in pregnant women with diabetes mellitus (DM) than in healthy pregnancies (3). Animal experimental studies have also shown that high blood glucose levels in early embryonic development tend to contribute to tail development malformation. The underlying defect is the failure of notochord formation during the gastrulation phase of embryogenesis. We describe a case of CRS in fetus of patient with pregestational diabetes (type 2) and discuss the ultrasound signs suggestive of CRS.
Case presentation
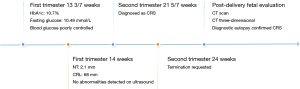
A 37-year-old woman, G3P1L1, was admitted to antenatal care in her third pregnancy. Her pregnancy at the time was complicated by obesity [body mass index (BMI) 31.8 kg/m2], chronic hypertension controlled with labetalol, and pregestational type 2 DM. She was diagnosed with type 2 DM in 2012. She took 50 mg of acarbose orally tid, and her blood glucose level was not controlled well. She was treated with insulin, Byetta (10 µg bi), YamoLi (1 mg qd in the morning and 2 mg qd in the evening), and Glucophage (0.5 g qid), but her blood glucose was not controlled well. At first trimester (13 and 3/7 weeks of gestation), the glycated hemoglobin (HbA1c) level was 10.7%, fasting glucose was 10.49 mmol/L, fasting insulin was 25.05 µIU/mL, and homeostatic model assessment for insulin resistance (HOMA-IR) index was 11.91, indicating inadequate glycemic control. During her pregnancy, she received insulin injections and dietary management, with 4 units of insulin aspart being administered subcutaneously 5 minutes before each meal (totaling 12 units daily) and 48 units of Levemir being subcutaneously administered at bedtime.
Her first pregnancy was terminated at 24 weeks when anencephaly of the fetus was detected, and ultimately a female stillborn baby with anencephaly was delivered. No genetic testing was carried out, and she had no relevant family history. Her second pregnancy required tight DM type 2 control and resulted in term delivery of a healthy infant via cesarean section.
Ultrasound at 14 weeks’ gestation revealed a singleton fetus with a normal nuchal translucency (NT) of 2.1 mm, a crown-rump length (CRL) of 68 mm, and normal amniotic fluid volume. No abnormalities were found on ultrasound. The two-dimensional (2D) abdominal sonogram was performed on a Voluson E8 device (HE HealthCare, Lanzhou, China) with a 4 to 8-MHz convex transducer. At this time, her HbAlc level was 7.7%, and her fasting glucose level was 5.9 mmol/L.
At 21 and 5/7 weeks of gestation, a detailed anatomical survey demonstrated abnormalities such as a lack of vertebrae in the sacral and caudal segments (Figure 1) and a deformed pelvis with an asymmetrical iliac bones on both sides of the spine, with the left iliac bone being higher than the right one. The right kidney could not be assessed, and the adrenal gland appeared to be flattened. No fetal limb deformity was observed. On a Doppler examination, a single umbilical artery was found. The placenta and amniotic fluid were normal, and there was no ultrasonographic sign of other anomalies. With these findings, CRS was diagnosed, and no obvious abnormalities were found in other fetal structures.
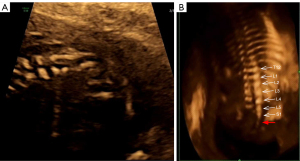
Following the diagnosis, the multidisciplinary team (including obstetricians, geneticists, and ethicists) provided the parents with a detailed discussion of CRS-associated outcomes based on current evidence. The parents were informed about the severity of the lesions and the resulting poor prognosis. They requested the termination of the pregnancy.
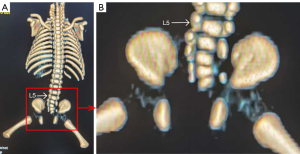
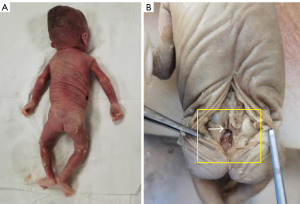
The pregnancy was terminated at 24 weeks of gestation, and the fetus was evaluated after delivery; the skin on the back of the fetus was intact, and a computed tomography (CT) scan of the stillborn fetus was performed. The findings were consistent with the absence of a partial caudal vertebra on prenatal ultrasonography. CT three-dimensional reconstruction could better show the absence of the coccyx with bilateral pelvic asymmetry (Figure 2). To better define the abnormalities of the spine, an autopsy was performed, and it was found that the end of the spine was abruptly terminated, and no corresponding vertebrae were present in the sacral or caudal regions (Figure 3). The key clinical events are summarized in Figure 4.
All procedures performed in this study were in accordance with the Declaration of Helsinki and its subsequent amendments. This study was approved by the Ethics Committee of the Gansu Provincial Maternity and Child Care Hospital (No. 2024GSFY07). Written informed consent was provided by the patient for publication of this article and accompanying images. A copy of the written consent is available for review by the editorial office of this journal.
Discussion
The primary cause of CRS is the disruption of the spinal cord complex during maturation before the 28th day of embryonic development, resulting in impaired development of the fetal sacrococcygeal spinal cord and spine (4). One study demonstrated that CRS is associated with uncontrolled maternal diabetes during pregnancy (5). Maternal diabetes may affect the metabolism of inositol and arachidonic acid and increase intracellular glucose levels, leading to a decrease in intracellular sodium concentration for embryonic tissue uptake. Both of these factors may cause disorders of embryonic disc coiling and affect the impaired closure of the hindgut and caudal neural tube. Therefore, CRS is often combined with abnormalities of the terminal spinal cord, anorectum, and genitourinary tract. Consequently, during the prenatal diagnosis of CRS, attention should be paid to the medical history of the pregnant woman to determine whether there is preexisting diabetes. In our case, the pregnant woman had type 2 DM for 11 years. At the first trimester (13 and 3/7 weeks of gestation), the HbAlc level was 10.7%, fasting glucose was 10.49 mmol/L, fasting insulin was 25.05 µIU/mL, and the HOMA-IR index was 11.91, indicating poor control. Preexisting maternal diabetes is a recognized risk for congenital malformations affecting various organ systems, although the exact mechanism is not fully understood. The relationship between maternal diabetes and fetal anomalies is well-documented, with numerous studies highlighting the increased risk of congenital malformations in the offspring of diabetic mothers. This association is particularly pronounced in cases where diabetes is poorly controlled before and during pregnancy. For instance, one study reported that an increased risk for congenital malformations is curvilinearly associated with higher HbA1C levels in early pregnancy among women with diabetes (6). Meanwhile, other research indicates that CRS also occurs in nondiabetic pregnant women, and it has been speculated that the disease is associated with inadequate blood perfusion and genetic susceptibility (7).
In early pregnancy, the diagnosis of CRS is difficult because the caudal vertebrae are not fully ossified; in our case, the CRL was smaller than normal for gestational week. Therefore, there should be heightened vigilance for this disease during early pregnancy if the CRL is smaller than normal for the gestational week, especially in cases of preexisting diabetes (8). The sole deviation from typical gestational development noted in this instance was a reduced fetal CRL, with no other structural abnormalities detected. This finding may be attributed to the possibility that the fetus is younger than the estimated gestational age. Furthermore, the pregnant woman’s elevated BMI could have adversely affected the quality of the ultrasound imaging, potentially obscuring existing structural abnormalities. In cases with the relevant medical history, a transvaginal examination may be considered in the future to enhance the early detection of fetal structural abnormalities.
CRS is most often diagnosed at midpregnancy, and the ultrasound presentation depends on the extent and severity of the spinal lesion. The typical sonogram shows a short abnormal spine in the sagittal view, with the absence of the caudal vertebrae (complete absence below S1 in this case); the iliac bones on both sides approaching at a smaller angle; the distance between the femoral heads shortened bilaterally; reduced movement of both lower limbs; and the posture fixed in a plank position. The prenatal identification of sacral agenesis and lower limb abnormalities prompted consideration of several differential diagnoses. First, open spina bifida was considered, which is typically associated with elevated maternal serum alpha-fetoprotein levels and characteristic lemon and banana signs on ultrasound; however, these indicators were not present in this case (9). Second, we considered VACTERL (vertebral defects, anal atresia, cardiac defects, tracheo-esophageal fistula, renal anomalies, and limb abnormalities), which requires the presence of at least three congenital anomalies (e.g., vertebral, anal, and cardiac defects) to be diagnosed. Although vertebral and renal anomalies were observed in our case, the absence of anal or cardiac abnormalities precluded this diagnosis (10). Ultimately, the combination of sacral agenesis and intact skin coverage over the spinal defect confirmed CRS as the definitive diagnosis (11).
The prognosis of CRS depends primarily on the degree of spinal cord end loss and the presence of other systemic abnormalities. Single caudal hypoplasia may be asymptomatic, but patients with vertebral and spinal cord hypoplasia or other concomitant deformities have a poor prognosis. The postnatal management of CRS requires a coordinated multidisciplinary strategy to effectively address the intricate medical and functional challenges associated with the condition. This strategy commences with early surgical planning, involving collaboration among pediatric orthopedic and urology specialists to address potential complications. Following surgical interventions, rehabilitation efforts prioritize the initiation of physical therapy during infancy to enhance motor function development. The overarching goal is to improve the quality of life and reduce the long-term burden of disability for affected individuals.
Acknowledgments
None.
Footnote
Funding: The study was supported by
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://qims.amegroups.com/article/view/10.21037/qims-24-2085/coif). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. All procedures performed in this study were in accordance with the Declaration of Helsinki and its subsequent amendments. This study was approved by the Ethics Committee of the Gansu Provincial Maternity and Child Care Hospital (No. 2024GSFY07). Written informed consent was provided by the patient for publication of this article and accompanying images. A copy of the written consent is available for review by the editorial office of this journal.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Sharma S, Sharma V, Awasthi B, Sehgal M, Singla DA. Sacral Agenesis with Neurogenic Bladder Dysfunction-A Case Report and Review of the Literature. J Clin Diagn Res 2015;9:RD08-9. [Crossref] [PubMed]
- Diel J, Ortiz O, Losada RA, Price DB, Hayt MW, Katz DS. The sacrum: pathologic spectrum, multimodality imaging, and subspecialty approach. Radiographics 2001;21:83-104. [Crossref] [PubMed]
- Aggarwal M, Sood V, Deswal S, Aggarwal KC. Caudal regression syndrome with bilateral popliteal webbing without maternal diabetes: a rare entity. Childs Nerv Syst 2012;28:1819-21. [Crossref] [PubMed]
- Isik Kaygusuz E, Kurek Eken M, Sivrikoz ON, Cetiner H. Sirenomelia: a review of embryogenic theories and discussion of the differences from caudal regression syndrome. J Matern Fetal Neonatal Med 2016;29:949-53. [Crossref] [PubMed]
- Bouchahda H, El Mhabrech H, Hamouda HB, Ghanmi S, Bouchahda R, Soua H. Prenatal diagnosis of caudal regression syndrome and omphalocele in a fetus of a diabetic mother. Pan Afr Med J 2017;27:128. [Crossref] [PubMed]
- Thorius IH, Petersen J, Husemoen LLN, Alibegovic AC, Gall MA, Damm P, Mathiesen ER. Glycemic Control and Risk of Congenital Malformations in Women With Type 1 Diabetes. Obstet Gynecol 2024;144:725-32. [Crossref] [PubMed]
- Mwamanenge N, Mariki H, Mkony M, Manji KP. Caudal regression syndrome without maternal diabetes mellitus. BMJ Case Rep 2023;16:e253136. [Crossref] [PubMed]
- Bashiri A, Sheizaf B, Burstein E, Landau D, Hershkovitz R, Mazor M. Three dimensional ultrasound diagnosis of caudal regression syndrome at 14 gestational weeks. Arch Gynecol Obstet 2009;280:505-7. [Crossref] [PubMed]
- Adzick NS, Thom EA, Spong CY, Brock JW 3rd, Burrows PK, Johnson MP, Howell LJ, Farrell JA, Dabrowiak ME, Sutton LN, Gupta N, Tulipan NB, D'Alton ME, Farmer DL. MOMS Investigators. A randomized trial of prenatal versus postnatal repair of myelomeningocele. N Engl J Med 2011;364:993-1004. [Crossref] [PubMed]
- Solomon BD. VACTERL/VATER Association. Orphanet J Rare Dis 2011;6:56. [Crossref] [PubMed]
- Jasiewicz B, Kacki W. Caudal Regression Syndrome-A Narrative Review: An Orthopedic Point of View. Children (Basel) 2023;10:589. [Crossref] [PubMed]


