
What role does artificial intelligence-driven quantitative analysis of chest computed tomography play in providing pulmonary function for idiopathic pulmonary fibrosis patients undergoing pirfenidone treatment?
Introduction
Idiopathic pulmonary fibrosis (IPF), the most severe and common idiopathic form of interstitial lung disease (ILD), carries a poor prognosis, with a 5-year survival rate of only 20–40% (1). However, in 2015, the U.S. Food and Drug Administration (FDA) approved two anti-fibrotic drugs—pirfenidone and nintedanib (2)—that have demonstrated significant efficacy in slowing disease progression and reducing acute exacerbations (3) in IPF patients. The Assessment of Pirfenidone to Confirm Efficacy and Safety in Idiopathic Pulmonary Fibrosis (ASCEND) trial showed that pirfenidone effectively reduced the incidence of disease progression, specifically in patients whose forced vital capacity (FVC) % predicted declined by more than 10% (2). Disease severity and response to treatment have been monitored in the clinic using pulmonary function tests (PFTs) such as FVC, the forced expiratory volume in one second divided by the FVC (FEV1/FVC) and/or diffusing capacity of the lung for carbon monoxide (DLCO). And American Thoracic Society (ATS)/European Respiratory Society (ERS)/Japanese Respiratory Society (JRS)/Latin American Thoracic Society (ALAT) guidelines (4) suggest that lung function is an important method for assessing the disease progression and predicting the disease prognosis in IPF.
High-resolution computed tomography (HRCT) is widely regarded as the most accurate non-invasive diagnostic tool for idiopathic ILD, particularly IPF (5). On HRCT, IPF typically presents with characteristic findings such as peripheral and basal predominant interstitial reticulation and honeycombing (1), which are crucial for diagnosis and monitoring disease progression (6,7). In clinical practice, serial HRCT scans, analyzed before and after pirfenidone, are often used to assess the degree of pulmonary fibrosis over time, helping inform timely adjustments to treatment plans that may reduce morbidity and mortality (8). However, several studies have shown substantial inter-observer variations in the visual (qualitative) evaluation of ILD by HRCT even among experienced thoracic radiologists (9-11). Visual scores are inconsistent and relatively irreproducible, and even experienced chest radiologists may struggle with the differential diagnosis. In this context, qualitative visual assessment can indicate whether lung parenchymal changes are present or not, but reliable quantification cannot be performed visually.
The importance of reliable measurement of fibrosis by HRCT is highlighted by data showing that systemic sclerosis patients with fibrosis progression >2% have a poorer prognosis (12). However, the monitoring of IPF remains reliant on the subjective visual assessment of fibrotic patterns on HRCT images (5). There is no universally accepted or standardized fibrosis scoring system, which introduces variability in the evaluation of disease progression. Recently, artificial intelligence (AI) quantification has been developing rapidly in ILD research, such as AIQCT, a quantitative CT model for predicting the prognosis of IPF (13), which automatically identifies and quantifies lung lesions and airway volumes through deep learning, providing additional prognostic information for IPF beyond the traditional gender, age, and physiology (GAP) staging (14). On the other hand, data-driven texture analysis quantifies the extent of fibrosis on HRCT using convolutional neural networks, which in turn generates a fibrotic score that correlates well with lung function and radiologists’ visual quantification of fibrosis, and can be used to characterize the extent of IPF and help predict disease progression (15,16). It automatically generates quantitative reports that provide an in-depth analysis of the distribution and severity of ILD. With the help of AI software solutions when reading chest CT in current day-to-day reading tasks, individual variations in radiology can be reduced (17).
In this study, we analyzed AI-driven quantitative analysis for long-term follow-up chest CT of IPF patients undergoing pirfenidone treatment. This study aimed to investigate the correlation between the fibrotic score derived from AI-driven quantitative analysis and PFT parameters in patients with IPF. Additionally, we sought to determine whether the fibrotic score could serve as a potential biomarker for monitoring the progression of pulmonary fibrosis in IPF patients. We present this article in accordance with the STROBE reporting checklist (available at https://qims.amegroups.com/article/view/10.21037/qims-2025-380/rc).
Methods
Study design
This retrospective study was conducted in accordance with the Declaration of Helsinki and its subsequent amendments. The study was approved by the Institutional Review Board of Jeonbuk National University Hospital (No. CUH 2024-02-003-004) and with a waiver of informed consent. Initially, 2,223 patients diagnosed with ILD via chest CT at Jeonbuk National University Hospital between January 2010 and July 2023, who had also undergone PFTs, were considered for inclusion. Patients who had not undergone surgical biopsy (n=1,990) or lacked PFT data (n=1) were excluded, leaving a final cohort of 232 patients. The inclusion criteria were as follows: (I) patients with typical or probable usual interstitial pneumonia (UIP) confirmed by both pathological diagnosis and chest CT, accompanied by PFT results; (II) patients with typical or probable UIP confirmed by pathology, but without UIP features on chest CT with high-resolution algorism, later confirmed as UIP following multidisciplinary discussion (MDD) with PFT results; and (III) patients who underwent AI-based analysis of the UIP pattern in both the initial and final follow-up CT with high-resolution algorism using AI software (Figure 1). Clinical data, including histological diagnosis, underlying conditions, pirfenidone, and PFT results, were retrieved from the electronic medical record. Among the 232 patients, we performed quantitative analysis of HRCT scans from 36 patients who survived for more than 10 years, regardless of pirfenidone treatment status, and who had both an initial HRCT and a follow-up HRCT performed after at least 10 years. The patients underwent HRCT scans once or twice per year, and a total of 684 serial CT scans were obtained from the 36 patients. Disease progression was assessed by comparing baseline and last follow-up CT scans to evaluate changes in response to pirfenidone over time.
CT examination
All CT scans were performed from the lung apices to the bases with patients in the supine position and full inspiration, using either a 64-channel CT scanner (Somatom Definition AS; Siemens Healthcare) or a 128-channel CT scanner (Somatom Definition Flash, Siemens Healthcare). CT parameters were as follows: 120 kVp; 60–90 mAs; the pitch of 0.75–1.5; collimation of 0.625–1.25 mm; sharp kernel with or without iterative reconstruction; 1-mm section thickness. Among the reconstruction parameters, sharp kernel (B60f), filtered back projection, and 1-mm-thick images were set as the representative images.
HRCT scans from 36 patients with UIP were first analyzed using an AI algorithm. A radiologist reviewed the AI-generated results. In cases where the radiologist did not agree with the AI analysis, a second thoracic radiologist (G.Y.J., with 23 years of experience) independently reviewed the findings, and discrepancies were resolved through revision of the initial interpretation. All images were analyzed on a workstation with optimized window settings for lung parenchyma assessment (window width: 1,500 HU; window level: −750 HU). IPF on CT was characterized by typical peripheral and basal interstitial reticulation and honeycombing, consistent with UIP, which correlated well with histopathological UIP patterns (18,19). Although IPF is inherently progressive, a ≥10% decline in FVC over 6–12 months is considered clinically significant and is often employed as a primary efficacy endpoint in drug trials, indicating a higher risk of mortality (20).
Deep learning-based texture analysis
Lung texture analysis was performed using commercially available deep learning-based software (Aview Lung Texture, version 1.1.43.7; Coreline Soft). This software utilizes a two-dimensional U-Net architecture and a deep convolutional neural network for fully automated segmentation and classification of lung parenchymal and ILD patterns (including emphysema, ground-glass opacity, solid changes, reticulation, and honeycombing) on CT images (19). Each HRCT slice was analyzed at the pixel level, yielding quantitative assessments of the relative volumes of six lung parenchymal patterns: honeycombing, reticulation, Ground-glass opacification, emphysema, solid lesions, and normal lung tissue. The whole lung volume was then spatially distributed at the pixel level by a validated content-based image retrieval system, dividing the whole lung volume into 64 rectangles (4×4×4), and the regional fraction of each disease pattern in each rectangle was calculated after extraction of the pattern (3).
The two best CT predictors of UIP are lower-lung honeycombing and upper-lung irregular lines. The finding of lower-lobe honeycombing was associated with a specificity of 69% for the diagnosis of UIP, compared with a specificity of 81% for the combination of lower-lung honeycombing with irregular lines in the upper lungs (21). Therefore, in this study, the fibrotic score was calculated as the sum of the mean percentages of the two fibrotic components—reticulation and honeycombing with/without ground-glass opacity base on these papers (22-24). The AI tools were applied to all image datasets, and readers were unaware of the final quantitative output of the automated texture analysis. Of the 89 CT scans in 36 patients, manual minor corrections were performed on 52 (58.4%) CT scans in 18 patients by examining each layer of CT images for each patient, correcting the range of reticular and honeycombing with/without ground-glass opacity images that were incorrectly identified by the AI, and re-calculating the mean percentage sum of the fibrotic scores using the AI to ensure that the fibrotic scores were accurately measured.
Statistical analysis
Different methods of representation were used according to the distributional characteristics of the clinical data and the HRCT categorical data. Specifically, continuous variables that conformed to normal distribution were expressed as mean ± standard deviation, whereas continuous variables that did not conform to normal distribution were expressed as median (interquartile range). Discontinuous variables were expressed as component ratios (%). The normality of all continuous variables was assessed using the Kolmogorov-Smirnov test or the Shapiro-Wilk test before parametric tests were performed, and the variance Chi-square was assessed using the Levene’s test. To compare the baseline characteristics and fibrotic scores between the oral pirfenidone-treated and non-treated groups, the Chi-squared test was used for categorical variables and the t-test for continuous variables. After initiating pirfenidone, patients were divided into two groups based on visual analysis: those showing little to no change and those with progression up to the final follow-up CT (25); patients who showed no or little change between the baseline and follow-up CT scans were classified as the “no or little change” group. All other patients were categorized as the “progression” group. Differences in fibrotic scores were then assessed between these groups. Continuous variables were assessed using independent samples t-tests. Correlations between FEV1/FVC, FVC, DLCO, and AI fibrotic scores were assessed using repeated measures analysis of variance (repeated measures ANOVA), with age-adjusted as a covariate. All analyses were conducted using IBM SPSS Statistics version 27 (IBM, Chicago, IL, USA), and statistical significance was defined as a P value of less than 0.05.
Results
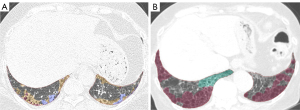
The final study population comprised 36 patients, with a mean age of 67.1±7.7 years. Of these, 21 (58.3%) were male and 15 (41.7%) were female. Seventeen patients (47.2%, mean age: 65.5±6.5 years) received pirfenidone, while 19 patients (52.8%, mean age: 68.6±8.6 years) did not. Twenty-five patients (69.4%) had underlying comorbidities, including 12 (33.3%) with hypertension, 12 (33.3%) with diabetes, and 10 (27.8%) with a history of cancer. A total of 6 (16.7%) patients died out of 36 patients, 2 (11.8%) out of 17 patients treated with pirfenidone, while 4 (21.1%) out of 19 patients were not treated with pirfenidone. Table 1 outlines the demographic and functional characteristics of this study, showing no statistically significant differences between those who received pirfenidone and those who did not (Figure 2).
Table 1
| Variables | Total (n=36) | Treatment of AFD (n=17, 47.2%) | No treatment of AFD (n=19, 52.8%) | P value |
|---|---|---|---|---|
| Age (years) | 67.1±7.7 | 65.5±6.5 | 68.6±8.6 | 0.225† |
| Sex | 0.535† | |||
| Male | 21 (58.3) | 9 (52.9) | 12 (63.2) | |
| Female | 15 (41.7) | 8 (47.1) | 7 (36.8) | |
| Underlying disease | 25 (69.4) | 14 (82.4) | 11 (57.9) | 0.112† |
| Hypertension | 12 (33.3) | 7 (41.2) | 5 (26.3) | 0.345† |
| Cancer | 10 (27.8) | 5 (29.4) | 5 (26.3) | 0.836† |
| Diabetes | 12 (33.3) | 7 (41.2) | 5 (26.3) | 0.345† |
| Pulmonary function test | ||||
| FEV1/FVC | 0.823‡ | |||
| Initial | 72.7±3.1 | 73.4±3.9 | 72.1±2.2 | 0.212† |
| Start AFD | 71.0±1.7 | 71.0±1.7 | – | – |
| Final | 70.2±1.9 | 70.3±1.7 | 70.1±2.3 | 0.845† |
| FVC (L) | 0.399‡ | |||
| Initial | 3.3±0.7 | 3.3±0.8 | 3.4±0.5 | 0.595† |
| Start AFD | 3.0±0.8 | 3.0±0.8 | – | – |
| Final | 2.8±0.7 | 2.8±0.9 | 2.9±0.6 | 0.552† |
| DLCO (%) | 0.968‡ | |||
| Initial | 81.0±22.9 | 83.8±27.7 | 78.5±17.9 | 0.827† |
| Start AFD | 61.7±17.5 | 61.7±17.5 | – | – |
| Final | 63.7±21.6 | 58.0±21.0 | 69.0±21.7 | 0.260† |
| AI fibrotic score (%) | 0.823‡ | |||
| Initial | 6.3±7.8 | 6.4±8.6 | 6.2±7.2 | 0.876† |
| Start AFD | 11.3±9.9 | 11.3±9.9 | – | – |
| Final | 19.6±16.1 | 16.6±12.5 | 22.3±18.7 | 0.471† |
Data are presented as n (%) or mean ± standard deviation. †, analyzed by t-test or Mann-Whitney U test (DLCO, AI fibrotic score); ‡, analyzed by repeated measure analysis of variance adjusted for age and sex, to evaluate the effect of treatment. AFD, antifibrotic drug; AI, artificial intelligence; DLCO, diffusing capacity of the lung for carbon monoxide; FEV1, forced expiratory volume in one second; FVC, forced vital capacity.
Comparative outcomes after pirfenidone
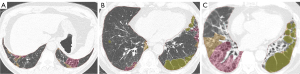
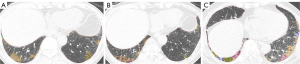
Table 2 compares the outcomes following pirfenidone. Among the 17 patients treated with anti-fibrotic agents, 11 patients (64.7%) demonstrated no or little change (Figure 3), while 6 patients (35.3%) exhibited progression (Figure 4). At the final follow-up, patients with no or little change (mean FVC: 2.8 L; mean DLCO: 64.1%) and those with progression (mean FVC: 2.6 L; mean DLCO: 47.3%) had lower post-treatment mean FVC and DLCO values compared to untreated patients (mean FVC: 2.9 L; mean DLCO: 69.0%). However, these differences did not reach statistical significance (P>0.05). In contrast, the mean progression rate of the AI fibrotic score was significantly higher in patients with progression after pirfenidone (mean AI fibrotic score, 27.1%) and in untreated patients (mean AI fibrotic score, 22.3%) compared to those with no or little change (mean AI fibrotic score, 10.9%) (P=0.006). Notably, in patients with IPF who demonstrated no or little change on HRCT following pirfenidone, a decline in DLCO (from 65.0%±18.9% to 64.1%±23.8%) and a reduction in the AI fibrotic score (from 12.6%±12.2% to 10.9%±8.7%) were observed at the final follow-up. Similarly, in cases showing progression on HRCT, a slight decrease in DLCO (from 52.5%±9.7% to 47.3%±9.4%) and a marked increase in the AI fibrotic score (from 12.8%±7.1% to 27.1%±12.1%) were noted at the final follow-up. These findings suggest that the AI fibrotic score may serve as a valuable tool for monitoring the therapeutic effects of pirfenidone in patients with IPF.
Table 2
| Variables | Not treatment of AFD (n=19, 52.7%) |
Treatment of AFD (n=17, 47.2%) | P value† | |
|---|---|---|---|---|
| No or little change after treatment of AFD (n=11, 64.7%) | Progression after treatment of AFD (n=6, 35.3%) | |||
| FEV1/FVC | ||||
| Initial | 72.1±2.2 | 73.9±4.7 | 72.5±1.6 | 0.311 |
| Start AFD | 71.0±1.8 | 71.0±1.5 | >0.99 | |
| Final | 70.1±2.3 | 70.3±1.9 | 70.2±1.5 | 0.979 |
| FVC (liters) | ||||
| Initial | 3.4±0.5 | 3.2±0.8 | 3.3±0.8 | 0.799 |
| Start AFD | 3.0±0.8 | 3.1±0.8 | 0.690 | |
| Final | 2.9±0.6 | 2.8±0.9 | 2.6±0.9 | 0.735 |
| DLCO (%) | ||||
| Initial | 78.5±17.9 | 88.8±32.5 | 74.7±13.8 | 0.383 |
| Start AFD | – | 65.0±18.9 | 52.5±9.7 | 0.235 |
| Final | 69.0±21.7 | 64.1±23.8 | 47.3±9.4 | 0.226 |
| AI fibrotic score (%) | ||||
| Initial | 6.2±7.2 | 7.0±10.4 | 5.3±4.3 | 0.915 |
| Start AFD | – | 12.6±12.2 | 12.8±7.1 | 0.969 |
| Final | 22.3±18.7 | 10.9±8.7 | 27.1±12.1 | 0.076 |
Data are presented as mean ± standard deviation. †, correlations between FEV1/FVC, FVC, DLCO, and AI fibrotic scores were assessed using repeated measures ANOVA (repeated measures ANOVA). AFD, antifibrotic drug; ANOVA, analysis of variance; AI, artificial intelligence; DLCO, diffusing capacity of the lung for carbon monoxide; FEV1, forced expiratory volume in one second; FVC, forced vital capacity.
Correlation between DLCO and AI fibrotic score in patients with IPF
A repeated measures ANOVA was conducted to assess the relationship between FEV1/FVC, FVC, DLCO, and the AI fibrotic score over time. The analysis revealed a significant interaction between these variables and time (F=13.695, P<0.001). The results demonstrated that DLCO declined over time, though the rate of decline slowed following pirfenidone. Additionally, the AI fibrotic score increased over time, in parallel with the DLCO decline, suggesting that the AI fibrotic score correlates with the worsening of diffusion capacity as pulmonary fibrosis progresses. This highlights the potential utility of the AI fibrotic score in assessing treatment response. However, changes in FEV1/FVC and FVC over time were not significant.
Discussion
This study demonstrated that utilizing AI to quantify HRCT in IPF patients undergoing pirfenidone could aid in monitoring treatment efficacy. In previous studies, physiological variants of PFT, particularly FVC and DLCO, have been reported as predictors of prognosis in IPF (4,26-30). In our study, however, AI-driven quantitative analysis of HRCT in IPF patients were consistent with changes in DLCO on PFT, thus allowing assessment of therapeutic response to pirfenidone. Most importantly, AI-driven quantitative fibrotic score on HRCT in IPF patients at long-term follow-up is associated with IPF progression.
Recent studies have reported the precision and reproducibility of CT-based fibrosis quantification, highlighting its potential for tracking disease progression (31,32). Ley et al. developed an enhanced GAP model (CT GAP model) that incorporates CT fibrosis scoring in place of DLCO (33). Lee et al. demonstrated that texture-based automated CT fibrosis quantification, particularly the fibrotic score, could serve as an independent predictor of survival in patients with IPF (34). Koh et al. reported that changes in CT quantification and FVC correlate with deep learning-based visual assessments of ILD progression (35). The results of Zhang et al. study showed that FEV1/FVC is used to assess airflow obstruction in COPD and correlates with emphysema parameters analyzed by CT quantitative analysis, whereas IPF usually manifests itself as a restrictive ventilatory disorder, which may be the reason why it did not correlate with CT quantitative indices (5). Lynch et al. found percent-predicted FVC was significantly inversely associated only with the extent of fibrotic score, but not with consensus diagnosis of IPF or presence of honeycombing (23). Jacob et al. found that quantitative, computer-derived CT metrics in IPF could serve as novel indices for IPF assessment (36). In our study, we used AI to evaluate fibrotic scores in HRCT scans of untreated IPF patients over a 10-year follow-up period. The analysis revealed approximately a fourfold increase in fibrosis progression compared to the initial CT scans. In cases without anti-fibrotic drug treatment, FVC and DLCO decrease by about 10% over ten years. However, changes in the fibrotic score did not correlate with the decline in lung function. This suggests that quantitative fibrotic progression on HRCT may not proportionally reflect declines in pulmonary function of untreated IPF patients in clinic.
In IPF patients, fibrotic score on quantitative CT may serve as a useful objective biomarker for monitoring purposes. Romei et al. reported significant differences in HRCT quantification progression between treated and untreated groups (37). Iwasawa et al. demonstrated that a computer-based quantitative CT analysis system exhibited substantial diagnostic power, with serial CT evaluations revealing a slow progression of pulmonary fibrosis following pirfenidone treatment, facilitating radiologists in assessing therapeutic response (38). In a Phase III-b placebo-controlled trial, Lancaster et al. showed that the increase in reticulation was significantly lower in the nintedanib group compared to the placebo group after 6 months, as measured by a quantitative texture analysis tool (39). Conversely, George et al. introduced the Weighted Vascular-Reticular Score, a novel metric quantifying combined reticular and vascular components from FVC-derived parameters, which can serve as a monitoring tool for treatment response in patients with fibrotic ILD (40). In our study, those who responded to pirfenidone showed stabilization or improvement in DLCO, along with either stable or reduced AI-derived fibrotic scores on HRCT. Conversely, in cases where DLCO continued to decline despite pirfenidone, the fibrotic score increased significantly. These findings suggest that the AI-derived fibrotic scores on HRCT can serve as a biomarker for monitoring the therapeutic efficacy of pirfenidone in IPF patients.
Despite the strengths of this study, several limitations should be noted. First, given the low prevalence of IPF in Asian populations and the frequently debated practice of withholding pirfenidone in functionally stable patients (41,42), and our cohort size remained relatively small even with over a decade of data collection. Consequently, these findings may not be fully generalizable to all IPF populations. Second, as a retrospective study, selection bias is a potential concern, and the imaging parameters (such as slice thickness and exposure settings) varied slightly across CT scans. However, Maldonado and Jacob have argued that these minor technical differences are unlikely to significantly impact the outcomes (43). Third, the interval between initiation of pirfenidone and outcome assessment was inconsistent across patients, ranging from 1 to 8 years. As a result, we were unable to evaluate the changes in CT findings at consistent intervals before and after the administration of pirfenidone. In future research, we aim to extend patient follow-up, integrate additional clinical data, and conduct a multicenter, large-sample study to explore the prognostic role of quantitative CT analysis in IPF and its applicability to other ILDs.
Conclusions
In summary, this study suggests that in patients with IPF, DLCO and AI-derived fibrotic scores on HRCT may serve as monitoring tools for assessing the therapeutic effects of pirfenidone. Improvement in DLCO following pirfenidone was associated with a reduction in AI-derived fibrotic scores. Given the limited number of patients surviving more than 10 years after an IPF diagnosis, these findings cannot be generalized. Therefore, further multicenter studies are warranted to evaluate whether AI-derived fibrotic scores from HRCT could serve as reliable biomarkers for monitoring disease progression or therapeutic response following pirfenidone in IPF patients.
Acknowledgments
None.
Footnote
Reporting Checklist: The authors have completed the STROBE reporting checklist. Available at https://qims.amegroups.com/article/view/10.21037/qims-2025-380/rc
Data Sharing Statement: Available at https://qims.amegroups.com/article/view/10.21037/qims-2025-380/dss
Funding: None.
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://qims.amegroups.com/article/view/10.21037/qims-2025-380/coif). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. This retrospective study was conducted in accordance with the Declaration of Helsinki and its subsequent amendments. The study was approved by the Institutional Review Board of Jeonbuk National University Hospital (No. CUH 2024-02-003-004) and with a waiver of informed consent.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Khan MA, Sherbini N, Alyami S, Al-Harbi A, Al-Ghamdi M, Alrajhi S, Rajendram R, Al-Jahdali H. Nintedanib and pirfenidone for idiopathic pulmonary fibrosis in King Abdulaziz Medical City, Riyadh: Real-life data. Ann Thorac Med 2023;18:45-51.
- Li H, Yang J, Chen S, Wang P, Yu X, Zhou Q, Zhang X, Zhang G. Analysis of the safety and efficacy of different plasma concentrations of pirfenidone in patients with idiopathic pulmonary fibrosis. Front Pharmacol 2022;13:1055702. [Crossref] [PubMed]
- Yang CC, Chen CY, Kuo YT, Ko CC, Wu WJ, Liang CH, Yun CH, Huang WM. Radiomics for the Prediction of Response to Antifibrotic Treatment in Patients with Idiopathic Pulmonary Fibrosis: A Pilot Study. Diagnostics (Basel) 2022.
- Raghu G, Remy-Jardin M, Richeldi L, Thomson CC, Inoue Y, Johkoh T, et al. Idiopathic Pulmonary Fibrosis (an Update) and Progressive Pulmonary Fibrosis in Adults: An Official ATS/ERS/JRS/ALAT Clinical Practice Guideline. Am J Respir Crit Care Med 2022;205:e18-47. [Crossref] [PubMed]
- Zhang H, Li X, Zhang X, Yuan Y, Zhao C, Zhang J. Quantitative CT analysis of idiopathic pulmonary fibrosis and correlation with lung function study. BMC Pulm Med 2024;24:437. [Crossref] [PubMed]
- Kwon BS, Choe J, Do KH, Hwang HS, Chae EJ, Song JW. Computed tomography patterns predict clinical course of idiopathic pulmonary fibrosis. Respir Res 2020;21:295. [Crossref] [PubMed]
- Chae KJ, Chung MJ, Jin GY, Song YJ, An AR, Choi H, Goo JM. Radiologic-pathologic correlation of interstitial lung abnormalities and predictors for progression and survival. Eur Radiol 2022;32:2713-23. [Crossref] [PubMed]
- Wang HY, Tsai SC, Lin YC, Hou JU, Chao CH. The effect of antifibrotic agents on acute respiratory failure in COVID-19 patients: a retrospective cohort study from TriNetX US collaborative networks. BMC Pulm Med 2024;24:160. [Crossref] [PubMed]
- Soffer S, Morgenthau AS, Shimon O, Barash Y, Konen E, Glicksberg BS, Klang E. Artificial Intelligence for Interstitial Lung Disease Analysis on Chest Computed Tomography: A Systematic Review. Acad Radiol 2022;29:S226-35. [Crossref] [PubMed]
- Walsh SL, Calandriello L, Sverzellati N, Wells AU, Hansell DM, Observer Consort UIP. Interobserver agreement for the ATS/ERS/JRS/ALAT criteria for a UIP pattern on CT. Thorax 2016;71:45-51. [Crossref] [PubMed]
- Widell J, Lidén M. Interobserver variability in high-resolution CT of the lungs. Eur J Radiol Open 2020;7:100228. [Crossref] [PubMed]
- Hoffmann T, Teichgräber U, Lassen-Schmidt B, Renz D, Brüheim LB, Krämer M, Oelzner P, Böttcher J, Güttler F, Wolf G, Pfeil A. Artificial intelligence-based quantification of pulmonary HRCT (AIqpHRCT) for the evaluation of interstitial lung disease in patients with inflammatory rheumatic diseases. Rheumatol Int 2024;44:2483-96. [Crossref] [PubMed]
- Handa T, Tanizawa K, Oguma T, Uozumi R, Watanabe K, Tanabe N, Niwamoto T, Shima H, Mori R, Nobashi TW, Sakamoto R, Kubo T, Kurosaki A, Kishi K, Nakamoto Y, Hirai T. Novel Artificial Intelligence-based Technology for Chest Computed Tomography Analysis of Idiopathic Pulmonary Fibrosis. Ann Am Thorac Soc 2022;19:399-406. [Crossref] [PubMed]
- Ley B, Ryerson CJ, Vittinghoff E, Ryu JH, Tomassetti S, Lee JS, Poletti V, Buccioli M, Elicker BM, Jones KD, King TE Jr, Collard HR. A multidimensional index and staging system for idiopathic pulmonary fibrosis. Ann Intern Med 2012;156:684-91. [Crossref] [PubMed]
- de la Orden Kett Morais SR, Felder FN, Walsh SLF. From pixels to prognosis: unlocking the potential of deep learning in fibrotic lung disease imaging analysis. Br J Radiol 2024;97:1517-25. [Crossref] [PubMed]
- Paik SH, Jin GY. Using Artificial Intelligence Software for Diagnosing Emphysema and Interstitial Lung Disease. J Korean Soc Radiol 2024;85:714-26. [Crossref] [PubMed]
- Jin GY. Preface for Special Issue on Clinical Experience of Artificial Intelligence for Thoracic Disease in Daily Practice. J Korean Soc Radiol 2024;85:691-2. [Crossref] [PubMed]
- Martinez FJ, Collard HR, Pardo A, Raghu G, Richeldi L, Selman M, Swigris JJ, Taniguchi H, Wells AU. Idiopathic pulmonary fibrosis. Nat Rev Dis Primers 2017;3:17074. [Crossref] [PubMed]
- Chae KJ, Lim S, Seo JB, Hwang HJ, Choi H, Lynch D, Jin GY. Interstitial Lung Abnormalities at CT in the Korean National Lung Cancer Screening Program: Prevalence and Deep Learning-based Texture Analysis. Radiology 2023;307:e222828. [Crossref] [PubMed]
- Dack E, Christe A, Fontanellaz M, Brigato L, Heverhagen JT, Peters AA, Huber AT, Hoppe H, Mougiakakou S, Ebner L. Artificial Intelligence and Interstitial Lung Disease: Diagnosis and Prognosis. Invest Radiol 2023;58:602-9. [Crossref] [PubMed]
- Hunninghake GW, Lynch DA, Galvin JR, Gross BH, Müller N, Schwartz DA, King TE Jr, Lynch JP 3rd, Hegele R, Waldron J, Colby TV, Hogg JC. Radiologic findings are strongly associated with a pathologic diagnosis of usual interstitial pneumonia. Chest 2003;124:1215-23. [Crossref] [PubMed]
- Shin KM, Lee KS, Chung MP, Han J, Bae YA, Kim TS, Chung MJ. Prognostic determinants among clinical, thin-section CT, and histopathologic findings for fibrotic idiopathic interstitial pneumonias: tertiary hospital study. Radiology 2008;249:328-37. [Crossref] [PubMed]
- Lynch DA, Godwin JD, Safrin S, Starko KM, Hormel P, Brown KK, Raghu G, King TE Jr, Bradford WZ, Schwartz DA, Richard Webb WIdiopathic Pulmonary Fibrosis Study Group. High-resolution computed tomography in idiopathic pulmonary fibrosis: diagnosis and prognosis. Am J Respir Crit Care Med 2005;172:488-93. [Crossref] [PubMed]
- Mogulkoc N, Brutsche MH, Bishop PW, Greaves SM, Horrocks AW, Egan JJGreater Manchester Pulmonary Fibrosis Consortium. Pulmonary function in idiopathic pulmonary fibrosis and referral for lung transplantation. Am J Respir Crit Care Med 2001;164:103-8. [Crossref] [PubMed]
- Aoki A, Hara Y, Fujii H, Murohashi K, Nagasawa R, Tagami Y, Enomoto T, Matsumoto Y, Masuda M, Watanabe K, Horita N, Kobayashi N, Kudo M, Ogura T, Kaneko T. The clinical impact of comorbidities among patients with idiopathic pulmonary fibrosis undergoing anti-fibrotic treatment: A multicenter retrospective observational study. PLoS One 2023;18:e0291489. [Crossref] [PubMed]
- Schwartz DA, Helmers RA, Galvin JR, Van Fossen DS, Frees KL, Dayton CS, Burmeister LF, Hunninghake GW. Determinants of survival in idiopathic pulmonary fibrosis. Am J Respir Crit Care Med 1994;149:450-4. [Crossref] [PubMed]
- King TE Jr, Safrin S, Starko KM, Brown KK, Noble PW, Raghu G, Schwartz DA. Analyses of efficacy end points in a controlled trial of interferon-gamma1b for idiopathic pulmonary fibrosis. Chest 2005;127:171-7. [Crossref] [PubMed]
- Paterniti MO, Bi Y, Rekić D, Wang Y, Karimi-Shah BA, Chowdhury BA. Acute Exacerbation and Decline in Forced Vital Capacity Are Associated with Increased Mortality in Idiopathic Pulmonary Fibrosis. Ann Am Thorac Soc 2017;14:1395-402. [Crossref] [PubMed]
- Hallstrand TS, Boitano LJ, Johnson WC, Spada CA, Hayes JG, Raghu G. The timed walk test as a measure of severity and survival in idiopathic pulmonary fibrosis. Eur Respir J 2005;25:96-103. [Crossref] [PubMed]
- Hamada K, Nagai S, Tanaka S, Handa T, Shigematsu M, Nagao T, Mishima M, Kitaichi M, Izumi T. Significance of pulmonary arterial pressure and diffusion capacity of the lung as prognosticator in patients with idiopathic pulmonary fibrosis. Chest 2007;131:650-6. [Crossref] [PubMed]
- Goldin JG, Kim GHJ, Tseng CH, Volkmann E, Furst D, Clements P, Brown M, Roth M, Khanna D, Tashkin DP. Longitudinal Changes in Quantitative Interstitial Lung Disease on Computed Tomography after Immunosuppression in the Scleroderma Lung Study II. Ann Am Thorac Soc 2018;15:1286-95. [Crossref] [PubMed]
- Walsh SLF, Calandriello L, Silva M, Sverzellati N. Deep learning for classifying fibrotic lung disease on high-resolution computed tomography: a case-cohort study. Lancet Respir Med 2018;6:837-45. [Crossref] [PubMed]
- Ley B, Elicker BM, Hartman TE, Ryerson CJ, Vittinghoff E, Ryu JH, Lee JS, Jones KD, Richeldi L, King TE Jr, Collard HR. Idiopathic pulmonary fibrosis: CT and risk of death. Radiology 2014;273:570-9. [Crossref] [PubMed]
- Lee SM, Seo JB, Oh SY, Kim TH, Song JW, Lee SM, Kim N. Prediction of survival by texture-based automated quantitative assessment of regional disease patterns on CT in idiopathic pulmonary fibrosis. Eur Radiol 2018;28:1293-300. [Crossref] [PubMed]
- Koh SY, Lee JH, Park H, Goo JM. Value of CT quantification in progressive fibrosing interstitial lung disease: a deep learning approach. Eur Radiol 2024;34:4195-205. [Crossref] [PubMed]
- Jacob J, Bartholmai BJ, Rajagopalan S, Kokosi M, Nair A, Karwoski R, Walsh SL, Wells AU, Hansell DM. Mortality prediction in idiopathic pulmonary fibrosis: evaluation of computer-based CT analysis with conventional severity measures. Eur Respir J 2017;49:1601011. [Crossref] [PubMed]
- Romei C, Tavanti LM, Taliani A, De Liperi A, Karwoski R, Celi A, Palla A, Bartholmai BJ, Falaschi F. Automated Computed Tomography analysis in the assessment of Idiopathic Pulmonary Fibrosis severity and progression. Eur J Radiol 2020;124:108852. [Crossref] [PubMed]
- Iwasawa T, Ogura T, Sakai F, Kanauchi T, Komagata T, Baba T, Gotoh T, Morita S, Yazawa T, Inoue T. CT analysis of the effect of pirfenidone in patients with idiopathic pulmonary fibrosis. Eur J Radiol 2014;83:32-8. [Crossref] [PubMed]
- Lancaster L, Goldin J, Trampisch M, Kim GH, Ilowite J, Homik L, Hotchkin DL, Kaye M, Ryerson CJ, Mogulkoc N, Conoscenti CS. Effects of Nintedanib on Quantitative Lung Fibrosis Score in Idiopathic Pulmonary Fibrosis. Open Respir Med J 2020;14:22-31. [Crossref] [PubMed]
- George PM, Rennison-Jones C, Benvenuti G, Sifostratoudaki A, Ottink FA, Bou-Zeid W, Ledda RE, Abul Kadir RF, Johari B, Fernandez C, Harston GWJ, Joly O, Gerry S, Devaraj A. Evaluation of e-Lung automated quantitative computed tomography biomarkers in idiopathic pulmonary fibrosis. ERJ Open Res 2024;10:e00570-2024. [Crossref] [PubMed]
- Behr J, Prasse A, Wirtz H, Koschel D, Pittrow D, Held M, et al. Survival and course of lung function in the presence or absence of antifibrotic treatment in patients with idiopathic pulmonary fibrosis: long-term results of the INSIGHTS-IPF registry. Eur Respir J 2020;56:1902279. [Crossref] [PubMed]
- Lederer DJ, Martinez FJ. Idiopathic Pulmonary Fibrosis. N Engl J Med 2018;378:1811-23. [Crossref] [PubMed]
- Jacob J, Bartholmai BJ, Rajagopalan S, Kokosi M, Egashira R, Brun AL, Nair A, Walsh SLF, Karwoski R, Wells AU. Serial automated quantitative CT analysis in idiopathic pulmonary fibrosis: functional correlations and comparison with changes in visual CT scores. Eur Radiol 2018;28:1318-27. [Crossref] [PubMed]


