
Prognostic value of cardiac magnetic resonance-derived myocardial segmental motion curve complexity metrics for recovery ability in post-myocardial infarction patients
Introduction
Myocardial infarction (MI), characterized by myocardial necrosis resulting from acute or prolonged coronary artery ischemia and hypoxia, represents a major threat to global human health due to its high mortality and morbidity rates (1,2). Despite the availability of aggressive therapeutic interventions, including thrombolytic therapy, percutaneous coronary intervention, and coronary artery bypass grafting, for patients with MI, the complete restoration of myocardial function after infarction remains a significant challenge. The ability of myocardial segments to heal significantly impacts subsequent patient care and the implementation of prognostic interventions. As a result, the timely and accurate assessment of myocardial recovery potential after treatment has emerged as a key challenge in the field.
Cardiac magnetic resonance (CMR) imaging and late gadolinium enhancement (LGE) imaging are diagnostic tools for MI. The former, characterized by its exceptional blood-tissue contrast and high reproducibility, remains the gold standard for monitoring myocardial features and cardiac function (3). Recently, myocardial strain features from cine CMR have been extensively employed for assessing myocardial motion, typically for evaluating dysfunction (4). Earlier studies described strong correlations between myocardial strain features, cardiac fibrosis, and conditions such as hypertrophic cardiomyopathy (5) and myocarditis (6). Furthermore, myocardial recovery potential in patients with MI has also been investigated by researchers through the utilization of myocardial strain features. Previous studies have established that myocardial strain characteristics provide independent or incremental predictive value for the prognosis of conditions such as MI (7), non-ischemic dilated cardiomyopathy (8), and myocarditis (9). Moreover, research reported that the presence and severity of LGE (10) and parameters such as the entropy of the peri-infarct border zone on LGE images (11) possess predictive potential for prognosis or adverse events in heart failure or MI patients.
As is well documented, complexity metrics (CM), which assess waveform randomness and disorder, play a decisive role in evaluating myocardial motion stability across directions, complementing myocardial strain assessments (12). This study further posits that CM may not be limited to revealing the current characteristics of myocardial fibrosis but could also reflect myocardial recovery capacity. Therefore, the basic hypothesis of this study is that CM parameters can uncover latent differences in motion characteristics between well-recovered myocardium and poorly recovered or deteriorating myocardium. It is anticipated that CM parameters, in combination with traditional segmental myocardial strain parameters, will enable the differentiation and evaluation of diverse states of myocardial recovery, thereby aiding clinicians in comprehensively evaluating treatment efficacy, tailoring personalized rehabilitation strategies, and effectively mitigating adverse consequences associated with poor MI prognosis. We present this article in accordance with the CLEAR reporting checklist (available at https://qims.amegroups.com/article/view/10.21037/qims-2024-2934/rc).
Methods
Study population
This retrospective cohort study included patients with MI who underwent paired CMR examinations, including both cine and LGE sequences. All enrolled patients completed the baseline CMR scan between November 2020 and November 2021 and the follow-up CMR scan between November 2021 and August 2022, with an interval of approximately one year between the two scans. Inclusion criteria were: (I) age between 18 and 90 years; (II) acute MI confirmation was based on European Society of Cardiology criteria (13), with concomitant fibrotic tissue evidence demonstrated via LGE sequences during initial imaging evaluation; (III) first-time acute MI diagnosis without prior MI or revascularization history. Exclusion criteria comprised: (I) individuals exhibiting significant functional limitation corresponding to New York Heart Association (NYHA) class III or IV; (II) estimated glomerular filtration rate (eGFR) <30 mL/min/1.73 m2; (III) concurrent non-ischemic cardiomyopathies or intracardiac devices (including atrial fibrillation rhythm disorders, rheumatic mitral stenosis, or mechanical valve prostheses); (IV) history of contrast media hypersensitivity; (V) contraindications to magnetic resonance (MR) scanning; (VI) microvascular ischemic diseases (e.g., MI with non-obstructive coronary arteries); (VII) history of confirmed prior MI (≥6-month history) or stable coronary artery disease (e.g., chronic coronary syndrome); (VIII) incomplete follow-up. All participants underwent emergency percutaneous coronary intervention with drug-eluting stent implantation as the primary reperfusion therapy. Ethical approval for this human subjects research was obtained from the Ethics Committee of Xuanwu Hospital (approval No. LYS2022169) following rigorous protocol review. This study was conducted in accordance with the Declaration of Helsinki and its subsequent amendments. Written informed consent was signed by all patients or their relatives.
This study systematically collected basic information on the subjects, as well as their history of cardiovascular disease and vascular risk factors. Assessment of vascular risk factors encompassed hypertension, diabetes, hypercholesterolemia, and history of smoking. (I) Hypertension was defined as having a blood pressure exceeding 140/90 mmHg or currently receiving antihypertensive drug therapy. (II) Hypercholesterolemia was determined by low-density lipoprotein cholesterol levels exceeding 140 mg/dL, high-density lipoprotein cholesterol levels below 40 mg/dL, triglyceride levels exceeding 150 mg/dL, or being treated with lipid-lowering medications. (III) Diabetes mellitus was diagnosed through fasting blood glucose ≥125 mg/dL, hemoglobin A1c (HbA1c) ≥6.5%, or current use of glucose-lowering medications. (IV) Individuals who had smoked within the year were categorized as smokers.
Imaging protocol
Paired CMR scans (baseline and follow-up) were acquired using an integrated PET-MR platform (uPMR 790, United Imaging Healthcare, Shanghai, China) with consistent imaging parameters. A 12-channel cardiac phased array coil was used for CMR imaging. During the scanning process, cine CMR sequences were employed to acquire images of myocardial motion in the left ventricular short-axis, four-chamber, and two-chamber views. High-quality images were obtained with the cooperation of subjects holding their breath. Only the left ventricular short-axis images were used for subsequent analysis in this study. Cine imaging was performed using a balanced steady-state free precession sequence without contrast administration. Key parameters included: repetition time/echo time =3.1/1.4 ms, flip angle 40°, acquisition matrix 288×256, and an acquisition time per slice of 10–15 s.
Following cine-CMR, LGE sequences were acquired. Contrast administration consisted of an intravenous gadolinium bolus (0.15 mmol/kg) injected at 2.0 mL/s using a power injector, followed by an equal volume normal saline. After a waiting period of 8–10 minutes, a phase-sensitive inversion recovery sequence scan was performed. The scanning planes and positioning for LGE were consistent with the cine CMR sequence. Parameters for the LGE sequence included: repetition time 4.7 ms, echo time 1.9 ms, flip angle 20°, matrix 240×180, and inversion time (TI) 300–330 ms.
From short-axis cine CMR images, the following left ventricular (LV) parameters were quantified: ejection fraction (%), end-diastolic volume index (mL/m2), end-systolic volume index (mL/m2), and LV mass (g/m2). The infarct size was defined as the percentage of infarct volume relative to the LV mass, which was calculated as the manually measured LGE scar volume as a percentage of the total LV volume.
Myocardial segmentation
The LV myocardium from cine CMR and LGE images were segmented using 3D Slicer (v5.22 stable release). Two trained researchers delineated the LV boundaries with minimal inter-operator variability. Following this, LV myocardial segmentation was performed by the criteria established by the Society for Cardiovascular Magnetic Resonance [2020] (3). Analysis exclusively incorporated cardiac imaging series demonstrating complete 360° myocardial circumferential visualization. Following the omission of apical myocardial regions, the left ventricle underwent standardized segmentation into basal, intermediate, and apical slices and further segmented into 6, 6, and 4 segments, respectively, totaling 16 myocardial segments according to guidelines. In the LGE images, MI areas were manually delineated by researchers and mapped to the corresponding 16 myocardial segments to calculate the transmural extension (TE) for each segment.
Category definition
In this study, myocardial segments were categorized based on their degrees of recovery using the following method. Firstly, the segment recovery rate (SRR, %) was determined by calculating the difference in myocardial TE between two scans.
where TE1 represents the TE at baseline, and TE2 denotes the TE at follow-up. Given that variations in the interval between patient follow-ups and scans could influence the magnitude of the SRR, the daily segment recovery rate (DSRR, ‰ per day), representing the average daily recovery, was calculated to eliminate the impact of intervals on myocardial recovery using the following formula:
where T stands for the interval (days) between scans. DSRR >0 reflects that the myocardial segment recovered (TE2<TE1), whereas DSRR ≤0 indicates that it did not recover or worsen.
Based on DSRR values, myocardial segments were classified into three categories according to the following criteria: myocardial segments that exhibited more than 50% recovery within 1 year (DSRR ×365 >50%, i.e., DSRR >0.14%) were defined as “High-recovery segments” (High segments), those that with evidence of recovery less than 50% within 1 year (0%< DSRR ≤0.14%) were defined as “Low-recovery segments” (Low segments), and myocardial segments that exhibited deterioration within 1 year (DSRR ≤0%) were defined as “Deteriorating segments” (Det segments).
Feature extraction
Strain, strain rate, and CM for individual myocardial segments were quantified at baseline using custom MATLAB algorithms (version 2018b, The MathWorks Inc, Natick, MA, USA) with 3D Slicer-generated myocardial masks. Myocardial segmental dynamics were analyzed through quantification of radial/circumferential dimensional changes using baseline cine-CMR datasets, enabling construction of tissue motion trajectories, which were then filtered using wavelet transformation, followed by feature extraction. The strain features (14), including radial/circumferential strain, strain rates, and CM features (12), encompassing frequency drift (FD), power spectral entropy, and permutation entropy, were extracted for each myocardial segment. Overall, each myocardial segment had four strain parameters and six CMs.
Classification
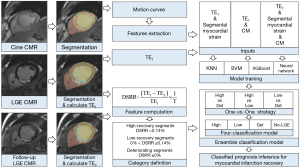
The current study aimed to classify myocardial recovery capacity using baseline characteristics, including MI severity (TE1), strain features, and CM features. The output was the DSRR category, which reflects the recovery degree. To train the classification model, ensemble methods employing the One-vs.-One strategy were utilized.
First, machine learning models were trained for one-on-one classification tasks: High vs. Det, High vs. Low, and Low vs. Det segments. To evaluate the prognostic efficacy of CMs, training was designed with three input sets: (I) TE1 + myocardial strain; (II) TE1 + CMs; (III) TE1 + myocardial strain + CMs. The performance of different input parameters in the same classification task was compared using the two-sided DeLong test to assess the importance of CMs in this step.
Then, the optimal classification model among the One-vs.-One models with different input parameters was selected. The outputs of these selected model were used as the inputs for a new training model, while the four-category classification of myocardium (High, Low, Det, and no-LGE) for 480 cases served as the model’s outputs for training. The integration of above models constituted the ensemble classification model for myocardial recovery capacity.
The classification models were trained using the K-nearest neighbor (KNN), non-linear support vector machine (SVM), eXtreme Gradient Boosting (XGBoost), and neural network (N-net) models. Class distribution imbalance was mitigated through implementation of cost-sensitive learning (15) combined with uniform under-sampling techniques. Five-fold cross-validation was implemented to ensure model robustness during iterative training. The general methodology for the model training process is shown in Figure 1.
Statistical analysis
Continuous parameters were quantified using mean values with standard deviations. Pearson correlation coefficients were calculated to investigate the correlations, with their corresponding P values indicating statistical significance. Univariate linear regression was performed, with β values validated using F-tests. Multivariate regression, with myocardial strain as a covariate, was employed to evaluate the predictive capability of CM. In the multivariate regression analysis, t-tests and variance inflation factor were used to assess significance and collinearity, respectively. Receiver operating characteristic (ROC) curves were plotted for each feature to distinguish among the three different degrees of MI, and the area under the curve (AUC) was calculated to evaluate their classification performance. Outliers were identified using the 3σ standard.
ROC curves or confusion matrix were plotted for the validation set of the trained KNN, SVM, XGBoost, and N-net models, and model classification efficacy was evaluated through performance metrics encompassing AUC, specificity, sensitivity, and accuracy quantification. All analyses were conducted using either Matlab or MedCalc (version 20.217, MedCalc Software Ltd, Ostend, Belgium). The classifiers were constructed using the Matlab Classification Toolbox (https://michem.unimib.it/download/matlab-toolboxes/classification-toolbox-for-matlab/).
Results
This study enrolled 30 MI patients, predominantly males (n=23), with an average age of 60.33±10.34 years. All participants underwent a follow-up exam approximately 1 year (329.3±66.1 days) post-baseline. Among 480 myocardial segments, 262 displayed LGE scars in the initial CMR: 99 High (37.8%), 102 Low (38.9%), and 61 Det (23.3%). The clinical traits and CMR metrics of individuals at baseline and follow-up are summarized in Table 1.
Table 1
| Characteristics | Value | |
|---|---|---|
| Baseline | Follow-up | |
| Baseline profiles | ||
| Age (years) | 60.33±10.34 | – |
| Sex (male) | 23 (76.67) | – |
| Diabetes mellitus | 12 (40.00) | – |
| Hypertension | 15 (50.00) | – |
| Dyslipidemia | 7 (23.33) | – |
| Smoker | 18 (60.00) | – |
| Heart rate (bpm) | 79.43±13.79 | – |
| Magnetic resonance parameters | ||
| Stroke volume (mL) | 70.48±17.05 | 73.81±13.88 |
| Ejection fraction of LV (%) | 50.07±11.95 | 52.63±10.38 |
| End-diastolic volume index of LV (mL/m2) | 82.39±20.08 | 82.03±23.05 |
| End-systolic volume index of LV (mL/m2) | 42.64±19.01 | 40.37±19.14 |
| LV mass (g/m2) | 107.2±29.12 | 100.9±23.56 |
| Infarct percent (% of LV mass) | 21.81±12.46 | 18.74±10.95 |
| Infarct location | ||
| Anterior | 7 (23.3) | – |
| Inferior | 16 (53.3) | – |
| Other | 7 (23.3) | – |
Continuous variables are expressed as mean ± standard deviation and categorical variables are presented as n (%). LV, left ventricular.
Correlation between myocardial features and DSRR
As presented in Table 2, correlations were investigated between segmental myocardial strain, CM, and the DSRR, which quantitatively assesses myocardial recovery capacity. The results revealed a significantly positive correlation between radial strain and DSRR (r=0.31, P<0.01), as well as between radial strain rate and DSRR (r=0.24, P<0.01). In contrast, the correlations between circumferential strain, strain rate, and DSRR were approximately zero, indicating no relationship.
Table 2
| Features | r† | β‡ | P§ |
|---|---|---|---|
| Segmental myocardial strain | |||
| Radial strain | 0.31** | 5.68×10−2** | |
| Radial strain rate | 0.24** | 3.01×10−2** | |
| Circumferential strain | 0 | – | |
| Circumferential strain rate | 0 | – | |
| Complexity metrics | |||
| Radial strain | |||
| Frequency drift | −0.33** | −8.61×10−4** | <0.05 |
| Power spectral entropy | −0.27** | −8.10×10−3** | 0.13 |
| Permutation entropy | −0.18** | −7.98×10−3** | 0.45 |
| Circumferential strain | |||
| Frequency drift | 0.15* | 3.88×10−4* | 0.14 |
| Power spectral entropy | 0.15* | 6.13×10−3* | 0.06 |
| Permutation entropy | −0.20** | −8.67×10−3** | <0.01 |
†, Pearson correlation coefficient. *, P<0.05; **, P<0.01. ‡, regression coefficients’ significance was F-tested. *, P<0.05; **, P<0.01. §, multiple linear regression with segmental myocardial strain covariates utilized t-test; collinearity assessment (VIF) demonstrated no significance (all <5). VIF, variance inflation factor.
Among the six CM extracted, three have a correlation coefficient greater than 0.2 with DSRR. The FD of the radial strain curve showed the strongest correlation with DSRR (r=−0.33, P<0.001), followed by the power spectral entropy of the radial strain curve (r=−0.27, P<0.001). Meanwhile, for circumferential strain-related CM, permutation entropy had the strongest correlation with DSRR (r=−0.20, P=0.002). Likewise, the FD of circumferential strain was significantly correlated with DSRR (r=0.15, P=0.016). The linear correlations between these four parameters and DSRR are depicted in Figure 2.
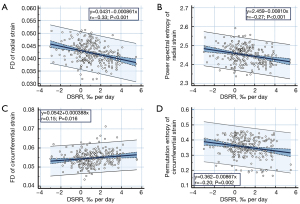
Univariate linear regression results (Table 2) showed regressive properties for most parameters except circumferential strain and strain rate (P<0.05). Furthermore, linear regression modeling was conducted incorporating myocardial segmental strain as covariates, to assess the independent predictive ability of CM features. Interestingly, the results uncovered no significant collinearity relationships among all parameters (variance inflation factor <5). Importantly, two key CMs remained significant independent predictors in the multivariate analysis (P<0.05). Taken together, these results highlight the relevance of CM features in assessing myocardial recovery.
Independent prognostic value of myocardial strain and CM
Moreover, the independent prognostic value of myocardial strain and CM was assessed. As listed in Table 3, when distinguishing myocardium with high recovery capacity from the remaining two categories, the independent predictive ability of the parameters was satisfactory (max AUC =0.73 for High vs. Det; max AUC =0.67 for High vs. Low). However, individual AUCs were low (max AUC =0.57) in differentiating Low from Det myocardium, indicating challenges in distinguishing these categories.
Table 3
| Features | AUC (95% CI) | ||
|---|---|---|---|
| High vs. Det | High vs. Low | Low vs. Det | |
| Segmental myocardial strain | |||
| Radial strain | 0.70 (0.62–0.77) | 0.65 (0.58–0.72) | 0.55 (0.47–0.63) |
| Radial strain rate | 0.65 (0.57–0.73) | 0.64 (0.56–0.70) | 0.53 (0.45–0.61) |
| Circumferential strain | 0.60 (0.52–0.68) | 0.58 (0.51–0.65) | 0.51 (0.43–0.59) |
| Circumferential strain rate | 0.54 (0.46–0.62) | 0.53 (0.46–0.60) | 0.50 (0.42–0.58) |
| Complexity metrics | |||
| Radial strain | |||
| Frequency drift | 0.73 (0.65–0.80)† | 0.67 (0.60–0.74)† | 0.55 (0.47–0.63) |
| Power spectral entropy | 0.65 (0.57–0.73) | 0.64 (0.57–0.71) | 0.52 (0.44–0.60) |
| Permutation entropy | 0.62 (0.54–0.70) | 0.57 (0.50–0.64) | 0.56 (0.47–0.64) |
| Circumferential strain | |||
| Frequency drift | 0.61 (0.53–0.69) | 0.67 (0.60–0.73) | 0.57 (0.49–0.65)† |
| Power spectral entropy | 0.62 (0.54–0.70) | 0.63 (0.56–0.70) | 0.53 (0.45–0.61) |
| Permutation entropy | 0.56 (0.48–0.64) | 0.60 (0.53–0.67) | 0.54 (0.46–0.62) |
†, the highest AUC value within each comparison group. AUC, area under the curve; CI, confidence interval; Det, deteriorating.
Different parameters exhibited differing prognostic values across the same classification task. For instance, the prognostic performance of CM was similar to or higher than that of myocardial strain features. Notably, the FD of radial/circumferential strain emerged as the most prominent feature (Figure 3). These findings collectively highlight the pivotal role of CM, particularly FD, in assessing myocardial prognosis.

Classification performance
This study utilized the One-vs.-One strategy to classify myocardial segments. Initially, three myocardial segment types were categorized using multiple algorithms, with myocardial strain parameters, CM, and their combination as inputs. Table 4 presents the classification results of various models in the validation set. The CM-input model exhibited favorable performance (AUC =0.700–0.822), outperforming the myocardial strain parameter model (AUC =0.662–0.795). As anticipated, the combination of both inputs further enhanced performance (AUC =0.708–0.826). DeLong test results indicated significant differences (P<0.05) in ROC curves between models using CM/combined inputs and those relying solely on myocardial strain parameters, suggesting distinct classification outcomes. Figure 4 demonstrates the ROC curves for the three optimal predictive models integrating myocardial strain parameters with CM. Indeed, these models could effectively differentiate between High vs. Det (AUC =0.826) and High vs. Low (AUC =0.802) but only moderately differentiate between Low and Det (AUC =0.708).
Table 4
| Models | High vs. Det | High vs. Low | Low vs. Det | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| AUC | Spec | Sens | ACC | AUC | Spec | Sens | ACC | AUC | Spec | Sens | ACC | |||
| Segmental myocardial strain | ||||||||||||||
| SVM | 0.785 | 71 | 70 | 71 | 0.795 | 74 | 77 | 75 | 0.662 | 69 | 61 | 67 | ||
| XGBoost | 0.767 | 78 | 61 | 72 | 0.785 | 72 | 79 | 74 | 0.651 | 61 | 65 | 61 | ||
| N-net | 0.782 | 79 | 60 | 72 | 0.790 | 72 | 74 | 73 | 0.609 | 62 | 63 | 62 | ||
| KNN | 0.794 | 75 | 75 | 75 | 0.778 | 71 | 78 | 73 | 0.552 | 66 | 57 | 64 | ||
| Complexity metrics | ||||||||||||||
| SVM | 0.770 | 73 | 75 | 74 | 0.792 | 75 | 75 | 75 | 0.657 | 61 | 70 | 63 | ||
| XGBoost | 0.822* | 77 | 83 | 79 | 0.772 | 76 | 71 | 74 | 0.654 | 62 | 67 | 63 | ||
| N-net | 0.758 | 71 | 75 | 72 | 0.779 | 76 | 78 | 77 | 0.589 | 59 | 67 | 61 | ||
| KNN | 0.782 | 79 | 72 | 76 | 0.798* | 74 | 78 | 76 | 0.700* | 66 | 67 | 66 | ||
| Segmental myocardial strain + complexity metrics | ||||||||||||||
| SVM | 0.782 | 78 | 74 | 76 | 0.798 | 74 | 84 | 77 | 0.654 | 61 | 70 | 62 | ||
| XGBoost | 0.826* | 82 | 81 | 82 | 0.781 | 71 | 75 | 72 | 0.619 | 59 | 61 | 60 | ||
| N-net | 0.752 | 70 | 77 | 72 | 0.798 | 72 | 74 | 73 | 0.630 | 63 | 61 | 62 | ||
| KNN | 0.778 | 71 | 81 | 74 | 0.802* | 73 | 80 | 76 | 0.708* | 66 | 70 | 67 | ||
Italic values indicate the highest AUC for each comparison group. *, demonstrated significant divergence from models incorporating segmental myocardial strain (P<0.05, DeLong test). ACC, accuracy; AUC, area under the curve; Det, deteriorating; KNN, K-nearest neighbor; N-net, neural network; SVM, support vector machine; XGBoost, eXtreme Gradient Boosting models.

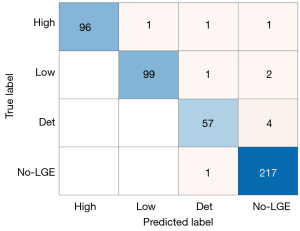
Subsequently, we utilized the output results of the One-vs.-One models as the input for a four-class classification model, with the myocardial segment categories (High, Low, Det, and no-LGE) serving as the output. The model was then trained accordingly. Figure 5 displays the confusion matrix of the four-class classification model trained using SVM. The model achieved an exceptionally high accuracy rate of 97.71%, indicating that this method is effective for classified prognosis inference in MI segmental recovery.
Discussion
This study, involving 30 MI patients, identified significant correlations between baseline CMR-derived CM parameters and myocardial recovery after one year. Of note, FD, a metric reflecting myocardial complexity, efficiently identified high-recovery myocardium. Moreover, machine learning models demonstrated that CM outperformed traditional myocardial strain parameters in prognostic efficacy. Moreover, the combination of CM with strain features further enhanced classification. These results collectively demonstrate the feasibility of predicting myocardial recovery using baseline features and highlight the crucial role of CM. Our findings imply that CM features are robust, reproducible imaging markers for evaluating myocardial recovery potential, thereby enhancing assessments in MI patients.
MI is a common myocardial necrotic condition with high mortality and morbidity. Post-treatment prognosis affects quality of life, and ~200,000 people in the US experience recurrent MI annually (16). While numerous studies have focused on early prognostic assessment of MI, this assessment provides not only an objective evaluation of treatment efficacy but also valuable guidance for subsequent treatment and rehabilitation strategies. Existing research has identified biomarkers (17) and features of electrocardiographic signals (18) as being closely related to the prognostic outcomes of MI. It is worth emphasizing that recent studies have further established the unique value of myocardial strain features for predicting the prognosis of patients with MI (7,19). As a series of features extracted from cine CMR, myocardial strain is recognized as an essential indicator for assessing myocardial motion status. Studies have identified a significant correlation between myocardial strain features and the degree of cardiac fibrosis, as well as strong associations with diseases such as myocarditis (6) and microvascular obstruction (20). According to previous studies, myocardial strain also possesses predictive utility for the prognosis of conditions such as MI (7,19), non-ischemic dilated cardiomyopathy (8), and myocarditis (9). This study posits that in addition to the motion amplitude characterized by myocardial strain, the stability of myocardial motion status characterized by CM may offer unique advantages in the context of diseases that alter myocardial properties, such as MI-induced myocardial fibrosis.
CM, a series of features used to assess the randomness and disorder of waveforms, is frequently employed in the field of mechanical engineering to evaluate the wear condition of mechanical equipment or identify faults. In recent years, CM parameters have also garnered considerable attention in the medical field. Regarding the key parameters mentioned in this study, FD has been widely utilized in previous research to assess neuronal oscillations (21), while power spectral entropy and permutation entropy are common measures of electroencephalogram complexity (22) used to calculate the randomness of signal spectra (23) or detect dynamic changes in time series (24). In cardiovascular imaging, researchers such as Zhao showed that the complexity of tissue edges holds clinical prognostic value (25). Recent studies have also unveiled that CM, including FD, can serve as feature parameters for assessing myocardial motion, exhibiting incremental performance in determining regional LGE volume in myocardial tissue (12).
This study identified a significant correlation between CM and the myocardial recovery capacity indicator DSRR. Specifically, FD, quantifying the temporal variability in periodic fluctuations of the motion curve (23), had a correlation coefficient of 0.33 with myocardial recovery proportion and exhibited superior prognostic performance in differentiating the three categories of myocardial recovery capacity compared to other parameters. Besides FD, both power spectral entropy and permutation entropy were correlated with myocardial recovery capacity (r>0.2). Furthermore, the combination of baseline parameters with CM as inputs for training the classification model markedly increased the classification performance compared to that of models solely using baseline parameters and traditional myocardial strain indices. When baseline parameters, myocardial strain, and CM were integrated as model inputs, the resulting ROC curve significantly differed from that of the model without CM parameters (DeLong test, P<0.05), yielding a superior prognostic performance.
The findings of this study validate the effectiveness of CM as a prognostic indicator for MI. In conjunction with recent research endeavors, CM not only characterizes and quantifies myocardial recovery capacity but also reflects the physiological state of the myocardium at the time of scanning. Although this study exclusively focused on three key indicators within the CM parameters, we postulate that a series of CM characteristics derived from myocardial motion curves have the potential to emerge as a significant category of features in the field of myocardial assessment, comparable to the role of radiomics in the evaluation of multiple diseases. We speculate that these CM features could provide robust support for the diagnosis and prognostic assessment of cardiomyopathies.
Based on baseline imaging and features in conjunction with an ensemble model employing the One-vs.-One strategy, this study achieved promising prognostic performance in assessing the recovery of myocardial segments with LGE. The adoption of the One-vs.-One strategy during training not only enhanced the efficiency of model training and data balance but also facilitated a precise understanding of the difficulty in classifying different types of myocardial segments, thereby laying the foundation for further improvements in future research. The results show that our One-vs.-One model demonstrated outstanding performance in distinguishing myocardial segments that recovered by more than 50% within one year from other types of myocardial segments (AUC =0.83, AUC =0.80). These results signal that even relying solely on baseline CMR images, our model can yield substantial and valuable clinical information regarding myocardial recovery capacity. However, regardless of myocardial strain parameters, CM, or the combination of both, the model faced challenges in differentiating between myocardial segments with low recovery capacity and those with deteriorating conditions (AUC =0.71), which may be addressed by integrating multimodal cardiac imaging or refining the classification of myocardial recovery levels.
This study derived a novel metric, DSRR, to assess myocardial recovery capacity based on a one-year follow-up dataset. DSRR provides an effective means of describing recovery status while accounting for data variability. The results of this study indicate that myocardial classifications based on DSRR can be effectively differentiated using myocardial strain and CM parameters. However, further studies are warranted to validate and elucidate its correlation with conventional clinical features.
Nevertheless, some limitations of this study cannot be overlooked. To begin, the primary limitation is the limited sample size stemming from challenges in the long-term follow-up of MI patients. Nonetheless, the conclusions and models are robust. Secondly, while this study innovatively introduced DSRR to evaluate myocardial recovery, its validation remains limited. Thirdly, manual segmentation limited CM feature application. Leveraging existing deep learning segmentation techniques for myocardial borders or large segmentation models could facilitate the automated analysis of cine CMR and subsequently enable a fully automated assessment of myocardial recovery capacity, providing additional information and efficacy for clinical practice. Lastly, only short-axis cine images were used, and further research on longitudinal myocardial strain CM characteristics is necessitated.
Conclusions
Herein, a significant correlation was observed between the complexity of myocardial segment motion curves, such as FD, power spectral entropy, and permutation entropy, and the recovery capacity of myocardial segments over a one-year period. Importantly, these CM parameters demonstrated relatively robust prognostic performance. As critical indicators of myocardial motion stability, these CM parameters could potentially serve as an effective tool for offering valuable and informative prognostic insights into the clinical assessment of myocardial physiological status.
Acknowledgments
The authors thank Jie Ma, Hongwei Yang, Dongmei Shuai and Lei Ma for the assistance with the patient studies.
Footnote
Reporting Checklist: The authors have completed the CLEAR reporting checklist. Available at https://qims.amegroups.com/article/view/10.21037/qims-2024-2934/rc
Data Sharing Statement: Available at https://qims.amegroups.com/article/view/10.21037/qims-2024-2934/dss
Funding: This work was supported by
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://qims.amegroups.com/article/view/10.21037/qims-2024-2934/coif). G.L. is supported by the 2024 Medical Engineering Research Project of National Institute of Hospital Administration (No. 2024MEA203). Y.H. is employed by Shanghai United Imaging Healthcare Co., Ltd. (a for-profit entity). This study received no financial or technical support from his employer or any commercial organization. The research conclusions are derived exclusively from standardized data analysis and do not assess proprietary software or products from any company, including his employer. Y.H.’s contributions to this work were strictly methodological and demonstrate no relation to his commercial affiliation or the interests of Shanghai United Imaging Healthcare Co., Ltd. Jie Lu is supported by the Huizhi Ascent Project of Xuanwu Hospital (No. HZ2021ZCLJ005). The other authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. Ethical approval for this human subjects research was obtained from the Ethics Committee of Xuanwu Hospital (approval No. LYS2022169) following rigorous protocol review. This study was conducted in accordance with the Declaration of Helsinki and its subsequent amendments. Written informed consent was signed by all patients or their relatives.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Nabel EG, Braunwald E. A tale of coronary artery disease and myocardial infarction. N Engl J Med 2012;366:54-63. [Crossref] [PubMed]
- Benjamin EJ, Muntner P, Alonso A, Bittencourt MS, Callaway CW, Carson AP, et al. Heart Disease and Stroke Statistics-2019 Update: A Report From the American Heart Association. Circulation 2019;139:e56-e528. [Crossref] [PubMed]
- Schulz-Menger J, Bluemke DA, Bremerich J, Flamm SD, Fogel MA, Friedrich MG, Kim RJ, von Knobelsdorff-Brenkenhoff F, Kramer CM, Pennell DJ, Plein S, Nagel E. Standardized image interpretation and post-processing in cardiovascular magnetic resonance - 2020 update: Society for Cardiovascular Magnetic Resonance (SCMR): Board of Trustees Task Force on Standardized Post-Processing. J Cardiovasc Magn Reson 2020;22:19. [Crossref] [PubMed]
- Tops LF, Delgado V, Marsan NA, Bax JJ. Myocardial strain to detect subtle left ventricular systolic dysfunction. Eur J Heart Fail 2017;19:307-13. [Crossref] [PubMed]
- Wang F, Xu X, Wang Q, Yu D, Lv L, Wang Q. Comparison of left ventricular global and segmental strain parameters by cardiovascular magnetic resonance tissue tracking in light-chain cardiac amyloidosis and hypertrophic cardiomyopathy. Quant Imaging Med Surg 2023;13:449-61. [Crossref] [PubMed]
- Gao Q, Yi W, Gao C, Qi T, Li L, Xie K, Zhao W, Chen W. Cardiac magnetic resonance feature tracking myocardial strain analysis in suspected acute myocarditis: diagnostic value and association with severity of myocardial injury. BMC Cardiovasc Disord 2023;23:162. [Crossref] [PubMed]
- Eitel I, Stiermaier T, Lange T, Rommel KP, Koschalka A, Kowallick JT, Lotz J, Kutty S, Gutberlet M, Hasenfuß G, Thiele H, Schuster A. Cardiac Magnetic Resonance Myocardial Feature Tracking for Optimized Prediction of Cardiovascular Events Following Myocardial Infarction. JACC Cardiovasc Imaging 2018;11:1433-44. [Crossref] [PubMed]
- Buss SJ, Breuninger K, Lehrke S, Voss A, Galuschky C, Lossnitzer D, Andre F, Ehlermann P, Franke J, Taeger T, Frankenstein L, Steen H, Meder B, Giannitsis E, Katus HA, Korosoglou G. Assessment of myocardial deformation with cardiac magnetic resonance strain imaging improves risk stratification in patients with dilated cardiomyopathy. Eur Heart J Cardiovasc Imaging 2015;16:307-15. [Crossref] [PubMed]
- Fischer K, Obrist SJ, Erne SA, Stark AW, Marggraf M, Kaneko K, Guensch DP, Huber AT, Greulich S, Aghayev A, Steigner M, Blankstein R, Kwong RY, Gräni C. Feature Tracking Myocardial Strain Incrementally Improves Prognostication in Myocarditis Beyond Traditional CMR Imaging Features. JACC Cardiovasc Imaging 2020;13:1891-901. [Crossref] [PubMed]
- Pi SH, Kim SM, Choi JO, Kim EK, Chang SA, Choe YH, Lee SC, Jeon ES. Prognostic value of myocardial strain and late gadolinium enhancement on cardiovascular magnetic resonance imaging in patients with idiopathic dilated cardiomyopathy with moderate to severely reduced ejection fraction. J Cardiovasc Magn Reson 2018;20:36. [Crossref] [PubMed]
- Wang L, Peng L, Zhao X, Ma Y, Jin F, Zhao X. Prognostic Value of Entropy Derived from Late Gadolinium Enhancement Images to Adverse Cardiac Events in Post-Myocardial Infarction Patients. Acad Radiol 2023;30:239-47. [Crossref] [PubMed]
- Li G, Zheng C, Cui Y, Si J, Yang Y, Li J, Lu J. Diagnostic efficacy of complexity metrics from cardiac MRI myocardial segmental motion curves in detecting late gadolinium enhancement in myocardial infarction patients. Heliyon 2024;10:e31889. [Crossref] [PubMed]
- Thygesen K, Alpert JS, Jaffe AS, Chaitman BR, Bax JJ, Morrow DA, White HD. Executive Group on behalf of the Joint European Society of Cardiology (ESC)/American College of Cardiology (ACC)/American Heart Association (AHA)/World Heart Federation (WHF) Task Force for the Universal Definition of Myocardial Infarction. Fourth Universal Definition of Myocardial Infarction (2018). J Am Coll Cardiol 2018;72:2231-64. [Crossref] [PubMed]
- Smiseth OA, Torp H, Opdahl A, Haugaa KH, Urheim S. Myocardial strain imaging: how useful is it in clinical decision making? Eur Heart J 2016;37:1196-207. [Crossref] [PubMed]
- Pes B, Lai G. Cost-sensitive learning strategies for high-dimensional and imbalanced data: a comparative study. PeerJ Comput Sci 2021;7:e832. [Crossref] [PubMed]
- Tsao CW, Aday AW, Almarzooq ZI, Anderson CAM, Arora P, Avery CL, et al. Heart Disease and Stroke Statistics-2023 Update: A Report From the American Heart Association. Circulation 2023;147:e93-e621. [Crossref] [PubMed]
- Kundi H, Balun A, Cicekcioglu H, Cetin M, Kiziltunc E, Topcuoglu C, Fevzi Kilinckaya M, Ornek E. Admission Value of Serum Cathepsin D Level Can be Useful for Predicting In-Hospital Mortality in Patients with NSTEMI. Acta Cardiol Sin 2017;33:393-400. [Crossref] [PubMed]
- Ando H, Yotsukura M, Sakata K, Yoshino H, Ishikawa K. Prognosis following acute myocardial infarction in patients with ST-T abnormalities on electrocardiograms obtained before myocardial infarction. Clin Cardiol 2001;24:107-13. [Crossref] [PubMed]
- Huang SJ, Ting I, Huang AM, Slama M, McLean AS. Longitudinal wall fractional shortening: an M-mode index based on mitral annular plane systolic excursion (MAPSE) that correlates and predicts left ventricular longitudinal strain (LVLS) in intensive care patients. Crit Care 2017;21:292. [Crossref] [PubMed]
- Gräni C, Stark AW, Fischer K, Fürholz M, Wahl A, Erne SA, Huber AT, Guensch DP, Vollenbroich R, Ruberti A, Dobner S, Heg D, Windecker S, Lanz J, Pilgrim T. Diagnostic performance of cardiac magnetic resonance segmental myocardial strain for detecting microvascular obstruction and late gadolinium enhancement in patients presenting after a ST-elevation myocardial infarction. Front Cardiovasc Med 2022;9:909204. [Crossref] [PubMed]
- Moca VV, Nikolic D, Singer W, Mureşan RC. Membrane resonance enables stable and robust gamma oscillations. Cereb Cortex 2014;24:119-42. [Crossref] [PubMed]
- Li R, Wu Q, Liu J, Wu Q, Li C, Zhao Q. Monitoring Depth of Anesthesia Based on Hybrid Features and Recurrent Neural Network. Front Neurosci 2020;14:26. [Crossref] [PubMed]
- Inouye T, Shinosaki K, Sakamoto H, Toi S, Ukai S, Iyama A, Katsuda Y, Hirano M. Quantification of EEG irregularity by use of the entropy of the power spectrum. Electroencephalogr Clin Neurophysiol 1991;79:204-10. [Crossref] [PubMed]
- Bandt C, Pompe B. Permutation entropy: a natural complexity measure for time series. Phys Rev Lett 2002;88:174102. [Crossref] [PubMed]
- Zhao X, Zhao X, Jin F, Wang L, Zhang L. Prognostic Value of Cardiac-MRI Scar Heterogeneity Combined With Left Ventricular Strain in Patients With Myocardial Infarction. J Magn Reson Imaging 2023;58:466-76. [Crossref] [PubMed]


